Enzymes
240 likes | 439 Vues
Enzymes. Characteristics. All Enzymes are Proteins Catalysts – i.e. control the rate of a chemical reaction. How Enzymes work. Enzymes bind and hold substrates (aka reactants ) in a certain orientation to speed the chemical reaction along Enzymes change shape as they bind the substrates.

Enzymes
E N D
Presentation Transcript
Characteristics • All Enzymes are Proteins • Catalysts – i.e. control the rate of a chemical reaction
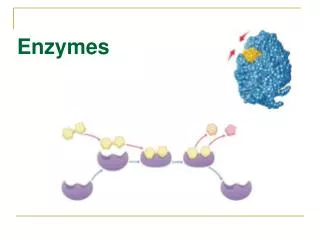
How Enzymes work • Enzymes bind and hold substrates (aka reactants) in a certain orientation to speed the chemical reaction along • Enzymes change shape as they bind the substrates
enzyme-substrate complex substrates the binding + active site
the reaction, the release enzyme-substrate complex product
What about the other way? substrate enzyme-substrate complex enzyme product
Lactase • 1926 aa’s long • cell membranes - small intestines Lactase
Beano - alpha galactosidase • breaks down trisaccharides • raffinose – in beans, cabbage • enzyme • not in humans • in bacteria in large intestines +
activation energy is the energy required to get a reaction going re-introducing activation energy activation energy net energy change
How do Enzymes work? • activation energy is the energy required to get a reaction going • They lower the “activation energy” of the reaction activation energy net energy change
How do they do it? • They lower the “activation energy” of the reaction net energy change
Enzyme performance is affected by: • amount of substrate present • temperature • pH • Inhibitors • Poisons
Enzymes and Amount of Reactants • [ reactants ] reaction rate because increased chance of finding molecules • [reactants] reaction rate because decreased chance of finding molecules 1. Measure [S] or [P] 2. Combine and Plot Experiment with amount and rate
Enzymes and Temperature • temperature reaction rate because increased kinetic energy breaks H-bonds • temperature reaction rate because decreased kinetic energy does not break H-bonds
pH • Acids – excess Hydrogen ions • Bases – excess hydroxyl ions • Neutral – equal numbers of H+ and OH-
Enzymes and pH • pHchanges reaction rate because H-bonds are altered • pHchanges reaction rate because H-bonds are altered Experiment with pH and rate
Enzymes and pH each enzyme has an optimal pH; some work best in acidic conditions (<4) (pepsin) while others work best closer to a neutral pH (7) pH for Optimum Activity Enzyme pH Optimum Lipase (pancreas) 8.0 Lipase (stomach) 4.0 - 5.0 Lipase (castor oil) 4.7 Pepsin 1.5 - 1.6 Trypsin 7.8 - 8.7 Urease 7.0 Invertase 4.5 Maltase 6.1 - 6.8 Amylase (pancreas) 6.7 - 7.0 Amylase (malt) 4.6 - 5.2 Catalase 7.0
Enzymes and Inhibitors bind to specific enzymes and decrease the reaction rate Noncompetitive inhibitor binds to the enzyme and changes its shape Normal substrate enzyme binding Competitive inhibitor binds to the active site
Poisons - KCN • Specific Irreversible Inhibitor of Cytochrome C Oxidase, • ATP cannot be made • Anaerobic respiration only • Fatal build up - Lactic Acid
Poisons - Arsenic • Nonspecific Inhibitor of cellular respiration enzymes • Inhibits glucose break down • Cell death results