Agarose Gel Electrophoresis
340 likes | 811 Vues
Agarose Gel Electrophoresis. What does gel electrophoresis do?. employs electromotive force to move molecules through a porous gel separates molecules from each other on the basis of size and/or charge and/or shape basis of separation depends on how the sample and gel are prepared.

Agarose Gel Electrophoresis
E N D
Presentation Transcript
What does gel electrophoresis do? • employs electromotive force to move molecules through a porous gel • separates molecules from each other on the basis of • size and/or • charge and/or • shape • basis of separation depends on how the sample and gel are prepared
Why perform electrophoresis on ds DNA? • To separate fragments from each other • To determine the sizes of fragments • To determine the presence or amount of DNA • To analyze restriction digestion products
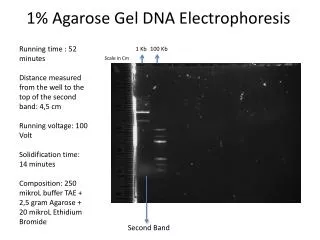
Length standards Ethidium bromide stained gel photographed on UV light box in black and white
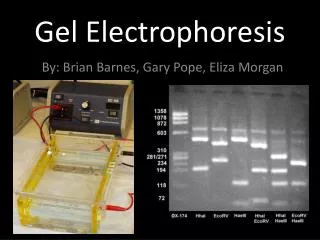
power supply gel box
Where does the current come from? • A direct current power supply • Ions supplied by the buffer • The charge on the macromolecules being separated • Electrolysis of water
Where does the current come from? • Electrolysis of water • 4H2O 2H2 + O2 + 2H2O • self-ionization of water throughout the buffer: 4H20 4H+ + 4OH- • At the negative pole • 4H+ + 4e- 2H2 • At the positive pole • 4OH- O2 + 2H2O + 4e- • At which electrode would you expect more bubbles? Why?
Basics of Gel Electric Circuits • V (volts) = I (milliamps) X R (resistance) • For a segment of a gel/buffer system • cross-sectional area of buffer or gel, resistance • strength of buffer = [ion], resistance • most resistance is in the agarose gel itself
What factors affect mobility of linear ds DNA? • Pore size of the gel • [agarose] pore size • pore size friction mobility • Voltage across the gel • voltage mobility • Length of the DNA molecule • smaller molecules generate less friction and so move faster • Ethidium bromide (stain) intercalated into DNA • decreases charge to mass ratio and so decreases mobility
Why don’t charge and shape affect mobility of linear ds DNA? • all DNA molecules have an essentially identical charge to mass ratio • 1 negative charge/phosphate and 1phosphate/base, so . . . . • charge is directly proportional to length • different lengths have essentially identical rod shape • Note: shape does affect mobility of circular and/or single strand DNA or RNA
Visualization • Monitoring the progress of the electrophoresis • tracking dyes visible to naked eye during run • xylene cyanol (migrates with ~5.0 kb fragments) • bromphenol blue (migrates with fragments of a few hundred base pairs) • Orange G (migrates with fragments of ~50 bp) • but mobility of tracking dyes can vary substantially depending on agarose • concentration • type
Visualization • Locating the DNA fragments in the gel • ethidium bromide staining • mutagen, wear gloves! • visible under UV light • wear UV opaque face or eye shield to observe!
Locating the DNA fragments in the gel • comparison to length standards Length standards Ethidium bromide stained gel photographed on UV light box in black and white
Factors affecting resolution • Resolution = separation of fragments • The “higher” the resolution, the more space between fragments of similar, but different, lengths • Resolution is affected by • agarose type • agarose concentration • salt concentration of buffer or sample • amount of DNA loaded in the sample • voltage
What is agarose? • linear carbohydrate polymer extracted from seaweed , agarbiose • forms a porous matrix as it gels • shifts from random coil in solution to structure in which chains are bundled into double helices
What is agarose? (cont’d) • multiple types of agarose • Standard agarose - LE • Gels at 35-38oC; Melts at 90-95oC • Becomes opaque at high concentrations • Makes a fairly sturdy gel • Low melting agarose (NuSieve) • Gels at 35oC; Melts at 65oC • Often used to isolate DNA fragments from gel • Modified by hydroxyethylation to lower M.P. • Is relatively translucent at high concentrations • Makes a fragile gel • Intermediate forms or combinations of LE and NuSieve can provide sturdy, translucent gels at high agarose concentrations • Good for resolving smaller fragments
Resolution of ds linear DNA fragments in agarose gels % Agarose (w/v)Size Range (kb prs) for Optimal Separation 0.5 2-30 0.75 0.7-20 1.0 0.5-10 1.5 0.2-3 2.0 0.1-2 3.0 (Nu-Sieve) 0.07-1.5 4.0 (N-S) 0.04-0.9 5.0 (N-S) 0.03-0.6 6.0 (N-S) 0.01-0.4
0.7% 2.5% Effect of agarose concentration on linear DNA fragment resolution. The two lanes contain identical DNA samples.
4M 1M 0M Effect of salt concentration on resolution of fragments. Samples in all three lanes are identical except for [salt].
0.1 0.25 0.5 1.0 5.0 DNA Hind III fragments (g) Effect of sample DNA concentration on resolution.
Voltage • voltage, rate of migration • to increase the voltage • increase the setting on the power supply • increase the resistance • decrease the gel thickness • decrease the ion concentration • if voltage is too high, gel melts • as voltage is increased, large molecules migrate at a rate proportionally faster than small molecules, so • lower voltages are better for resolving large fragments • but the larger ds DNA fragments are always slower than the smaller ones
Buffer Systems • Remember, buffer systems include weak acids and/or bases that do not dissociate completely. • If ions resulting from dissociation are “removed,” more weak acid and/or base will dissociate. • Purposes of buffer • Keep solution at pH compatible with molecules being separated • Generate ions consistently to • maintain current • keep resistance low • Both gel and the solution in the gel box are buffered.
Buffer Systems (cont’d) • Two commonly used buffers for routine agarose gel electrophoresis • TAE, pH 8.0, ~50 mM - Tris, Acetate, EDTA • TBE, pH 8.0, ~50 mM - Tris, Borate, EDTA • Tris (T) is a weak base. • Acetic (A) acid and boric (B) acid are weak acids. • Acetic acid is more completely ionized at pH 8.0 than is boric acid, so TBE has a high buffer capacity than TAE.
Buffer Systems (cont’d) • TAE, pH 8.0, ~50 mM - Tris, Acetate, EDTA • loses buffer capacity during long or high voltage gel runs; • anode end of gel becomes acidic • gel may melt from the increased resistance that results from ion depletion • resolves high MW fragments better than TBE • TBE, pH 8.0, ~50 mM - Tris, Borate, EDTA • higher buffer capacity • somewhat more expensive • resolves low MW fragments better than TAE • may interfere with subsequent reactions
Non-denaturing agarose gel loading solutions • Composition • tracking dyes • are used to follow progress of electrophoresis • sometimes interfere with later visualization of DNA • a solute to increase density • so that sample falls to bottom of loading well with minimal dilution • solute examples: glycerol, Ficoll • Other gel types, with different purposes, use different loading solutions!
Ethidium bromide staining • Binds to DNA by intercalation between stacked bases • lies perpendicular to helical axis • makes Van der Waals contacts with bases above and below • + charge, • so migrates toward negative pole • reduces the charge to mass ratio of the DNA fragment to which it is bound • Because of the change in charge to mass ratio, it alters DNA mobility, especially of circular covalently closed DNA
Ethidium bromide staining • Used to visualize DNA with UV light • uv 254 nm absorbed by DNA and transmitted to EtBr • excitation at 302 or 366 nm fluorescence at 590 nm • 560 nm = visible red/orange • >/= 10ng/band required for visualization • Bound dye fluoresces 20-25X more than dye in solution because of • fixed position of planar group • proximity of dye to bases • UV light damages eyes and skin! Wear goggles and/or face shield.
Trouble shooting • Smearing • torn sample wells • voltage too high for large fragments • too much DNA • Use </= 0.5 ug / fragment / 0.25cm2 migration area • Gel melts • voltage too high • ionic strength too low • Poor resolution • wrong [agarose] • small bands are fuzzy – the gel run may have been too long at too low a voltage, allowing diffusion of the DNA and broadening of the band