Ventricular Septal Defects
Ventricular Septal Defects. 1 in 1000 live births 50% associated with other congenital malformations First described by Roger in 1879, hence small VSDs are also known as the ‘maladie de Roger’. First VSD closed under direct vision by Lillehei in 1955. Ventricular Septum. Components

Ventricular Septal Defects
E N D
Presentation Transcript
1 in 1000 live births • 50% associated with other congenital malformations • First described by Roger in 1879, hence small VSDs are also known as the ‘maladie de Roger’. • First VSD closed under direct vision by Lillehei in 1955
Ventricular Septum • Components • Membranous septum • Outlet or conal septum • Inlet septum or the septum of the A-V canal • Muscular or trabecular septum
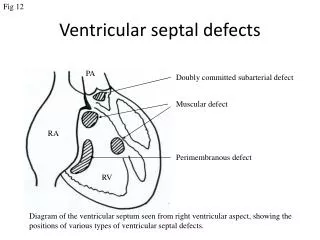
Anatomic Classification of VSDs • Perimembranous • Subarterial or outlet • A-V canal or Inlet • Muscular
Perimembranous VSD • Also called conoventricular defects. • Most common (80%) • Margins include membranous septum or remnant • May have extensions into inlet, outlet or trabecular septum • Postero-inferior margin very close to the antero-septal commissure of the Tricuspid valve • Can extend upto non-coronary cusp of aortic valve
Subarterial VSDs • Also k/a outlet, conal, subpulmonary, supracristal • Located in the outlet septum • 5 – 10% of VSDs • Can extend upto right or sometimes non-coronary cusps of the aortic valve, hence cause AR
A-V canal or Inlet VSDs • In the inlet part of the septum, usually spanning the length of the septal leaflet of the TV • < 5% of surgically treated VSDs • The posterior rim of the defect runs along the septal leaflet of the TV to the central fibrous body and the AML (which may have a cleft in association). • No intervening muscle between the VSD and the tricuspid valve
Muscular VSDs • Muscle tissue all around the defect • According to their location they may be either anterior, in the inlet septum, mid-muscular or apical • Classification according to location is important because it determines the approach for surgical closure. • Inlet and mid-muscular ----- RA approach • Anterior ------- Rt. Ventriculotomy • Apical ------ May require left ventriculotomy • May be multiple
Atrioventricular conduction system • AV-node -------- Triangle of Koch • Bundle of His ------ through CFB in close relation to both the tricuspid and aortic valves • Along inferior border of the membranous septum • Left ventricular side of muscular IVS • Perimembranous VSD ----- Conduction tissue along postero-inferior rim of the defect • Inlet VSD ----- AV node migrates posteriorly towards the crux of the heart, hence the conduction tissue lies along the postero-inferior margin again • Subarterial VSD ---- Muscle tissue between rim of VSD and conduction tissue • Muscular VSD ----- Conduction tissue is in normal anatomic position. Separated from rim by muscular tissue
Pathophysiology • Two determinants • Size of defect • Pulmonary vascular resistance • These determine • Pressure gradient across VSD • Shunt volume across VSD • After birth PVR falls ------ Large flow across shunt if large VSD • Causes increased PA pressure (initially flow related), increased PV return, hence LA enlargement and LV overload • PH initially flow related and reversible
Pathophysiology • Later ----- Intimal proliferation and medial hypertrophy leads to fixed irreversible PH • Flow through the lungs decreases as PVR increases, hence shunt volume decreases • Eventually PVR > SVR, hence R L shunt across VSD Cyanosis Eisenmengerised VSD • Shunt calculated by Fick’s principle Qp/Qs Aortic O2 % sat - Central Venous O2 % sat Pulm. Vein O2 % sat – Pulm. Art O2 % sat • With small VSDs, there is resistance to flow across the VSD hence Qp/Qs is rarely > 1.5 • With moderate VSDs, Qp/Qs is between 1.5 and 2.5, and is less likely to cause pulm vasc disease
Symptoms & Natural History • Both depend on shunt volume • With large VSDs, infants present with respiratory infections, failure to thrive and heart failure. • If untreated, 10% will die within first year of life • If they survive beyond the first year, if untreated, with eventually develop Eisenmenger complex and die (Hemoptysis, polycythemia, cerebral abscesses and infarction and right heart failure) • Smaller VSDs produce less symptoms, and survival to older ages is known • Bacterial endocarditis risk 0.15 to 0.3 % per year
Symptoms & Natural History • Spontaneous closure is known, primarily with perimembranous and muscular VSDs. Subarterial and inlet VSDs rarely • Chances differ with age at detection • At 1 month 80% of large VSDs close • At 6 months 50% • At 12 months 25% • AI – with subarterial VSDs or Perimembranous VSDs with subaortic extensions
Clinical features • Depends on shunt flow • Spectrum of symptoms • Small VSDs rarely symptomatic • Large VSDs with severe symptoms failure to thrive, heart failure, repeated LRTI • Hyperactive precordium • Pansystolic precordial murmur • Signs may change with spontaneous closure of Eisenmengerisation
Diagnosis • ECG - Left atrial enlargement, ventricular hypertrophy • CXR – Cardiomegaly d/t LA enlargement and LV volume overload • Increased vascular markings (plethoric lung fields) • 2D, Doppler Echo and CF mapping • Cardiac Cath • Not routinely performed • To be considered in older patients to document PA pressure and PVR • Can also calculate Qp/Qs, and irreversible component of PH
Treatment • Treatment dictated by • Severity of symptoms • Age at presentation • PVR • Size and type of defect • Other co-existing malformations • Early repair in infancy for intractable HF and failure to thrive • With less symptoms, best to wait till 12 months of age for spontaneous closure • Thereafter, moderate to large VSDs should be closed
Treatment • Infants > 6 months, with raised PVR – prompt surgery • Inlet and subarterial VSDs – early closure • Surgical approach • Standard CPB with bicaval cannulation and snaring of Cavae • In very small infants < 8 kg, circulatory arrest with deep hypothermia may be best • RA approach for most VSDs • Subarterial VSDs may be approached through PV • Muscular VSDs approach depends on location • Usually closed with Veloured Dacron or Goretex patches • Interrupted or continuous sutures
Treatment • Damage to conduction tissue avoided by • Placement of sutures a few mm away from the rim on the right ventricular side of the septum, in the region of proximity to the • Where there is no tissue between the rim of the VSD and the tricuspid valve, sutures are placed on the base of the tricuspid septal leaflet
Results • Early mortality approaching 0% in specialised centers • Patients operated late, with high PVR have worse long-term outcome • Conduction disturbances • CHB approaching 0% in specialised centers • RBBB 30 – 35% with RA approach, higher if RV approach • Residual defect 1 – 2%