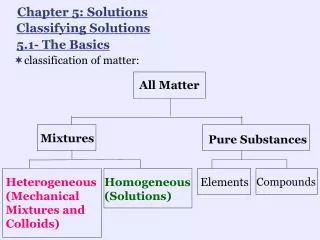
What’s the Matter?
What’s the Matter?. What’s the Matter?. Matter is the “stuff” that all objects and substances are made out of. What’s the Matter?. All matter takes up space (volume) All matter contains a certain amount of material (mass) All matter can be detected and measured. What’s the Matter?.

What’s the Matter?
E N D
Presentation Transcript
What’s the Matter? Matter is the “stuff” that all objects and substances are made out of
What’s the Matter? • All matter takes up space (volume) • All matter contains a certain amount of material (mass) • All matter can be detected and measured
What’s the Matter? • Some matter, such as bacteria or cells, is so small that you need a microscope to see it • Some matter, such as gas, you can only detect by smelling it • Some matter, such as air, you can only detect when you feel it on your skin or see it move other matter (such as leaves or flags)
Properties A property is a characteristic of matter that helps identify or classify it.
Physical Properties Physical Properties can be observed without changing the composition or identity of the matter.
Examples of Physical Properties • Density – amount of matter in a given volume (density = mass/volume) • Ductility – ability to be pulled into wire • Malleability – ability to be pressed or pounded into thin sheets
Examples of Physical Properties • Boiling Point – temperature where substance changes from a liquid to a gas • Melting Point – temperature where substance changes from a solid to a liquid
Examples of Physical Properties • Electrical Conductivity – how well a substance allows electricity to flow through it • Solubility – ability to dissolve into another substance
Chemical Properties Chemical Properties describe matter based on its ability to change into a new kind of matter with different properties.
Examples of Chemical Properties • Flammability – how easily something burns • Rusting – iron reacts with oxygen to form rust • Reactivity – how substances react with water, acid and other substances