Amino Acids
160 likes | 412 Vues
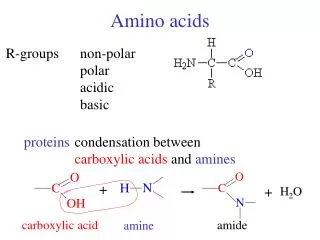
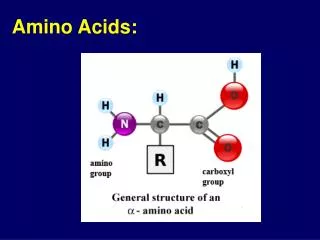
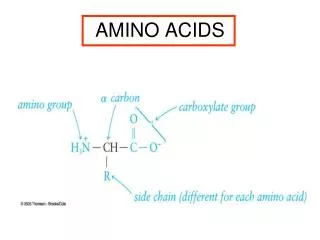
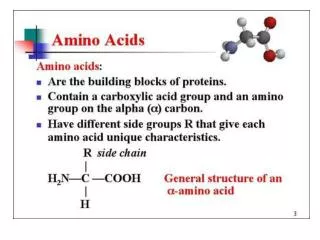
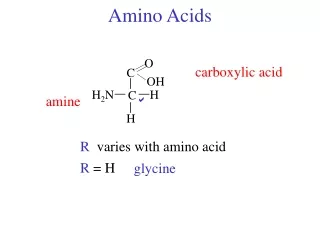
Amino Acids. Amino Acid Structure. Know the parts of an amino acid 1. Amino group 2. Carboxylic Acid (Carboxylate) 3. R-Group. R-Groups. Amino acids are classified by their R-groups and the groups interaction with water 1.Hydrophobic-The majority of R-groups 2. Polar Uncharged

Amino Acids
E N D
Presentation Transcript
Amino Acid Structure • Know the parts of an amino acid • 1. Amino group • 2. Carboxylic Acid (Carboxylate) • 3. R-Group
R-Groups • Amino acids are classified by their R-groups and the groups interaction with water • 1.Hydrophobic-The majority of R-groups • 2. Polar Uncharged • 3. Polar Charged
Hydrophobic R-Groups • R-groups composed of only Hydrogen and Carbons • One exception- Methionine contains sulfur, bound only to Carbon Phenylalanine Methionine
Polar uncharged and charged • Look for Oxygens and Nitrogens • Charged groups will be charged or have a primary amine or carboxylic acid part. • Primary amines are written NH2 and Carboxylic acids COOH
Acid Base Properties • Amino acids can be proton donors • The pH at which the Carboxylate group gives up its proton is the pK1 and the Amino group is pK2 • pK1 is the point where 50% of the carboxylate groups are unprotonated, and pK2 is where 50% of the amino groups are unprotonated • Amino acids are good buffers at these points • Add them together and divide by 2 to get the isoelectric point (pI) • This is the point where all amino acids in a solution have zero net charge • Amino acids are bad buffers at this point
Amino acids can form polypeptides • Peptide Linkages: C-N bond • Different order equals different peptides • Ex: Gly-Ala-Pro isn’t the same as Pro-Ala-Gly • Number of possible combinations using each AA once: (Number of Amino Acid)! • Ex: tetrapeptide is 4! (4x3x2x1=24) • Number of possible combinations using each AA as many times as we want: (number of AA)number in polypeptide • Ex: 4 amino acids in a tetrapeptide =44
Amide planes • An amide plane forms around the peptide linkage • C,N,O,H are involved in the formation • The double bonded O electrons resonate to the C-N bond, giving the peptide linkage a partial double bonded characteristic.
Protein Substructures • Primary • Order of the amino acids • Ex: ala-gly-asp-leu-lys-phe • Main type of interaction: covalent between amino acids, called peptide bond • Secondary • Alpha helix • Main type of interaction: hydrogen bonds between amide planes
Protein substructure • Tertiary • Main type of interactions: between R-groups • H-bonds, Electrostatic, Hydrophobic, Covalent (disulfide bonds) • Specifically, covalent bond called disulfide • Between 2 cysteines’ sulfhydryl R-groups • Quaternary • Large molecular weights
Collagen • Predominate AA: proline and glycine • Gly- small R-group for rope-like structure • Pro- Oxidizable R-group • Requires Vitamin C; deficiency=scurvy • Steps in formation • Immature collagen is hydroxylated (-OH groups added) becoming procollagen • Need Vitamin C as reducing agent in this step. If deficient, the person has scurvy • Procollagen is glycolyslated (carbohydrate moeities added) making tropocollagen • Mature collagen formed by crosslinks between hydroxylated prolines
Hemoglobin and Gas Exchange • Bohr Effect
Gas exchange driven by pH • HHb (protonated Hemoglobin) enters lungs • Lungs are more basic, so HHb more likely to give up proton • Oxygen (O2) binds to hemoglobin and it gives up the H+ • HHb+ O2+HCO3- HbO2+H2O+CO2 • The bicarbonate is the form that carbon dioxide goes to the lungs as. It travels in the blood plasma. • All of the reactions occur inside the red blood cell
Gas Exchange • Oxygenated hemoglobin travels to the tissues where it encounters an acidic enviroment. • The acidity is the result of increase carbon dioxide, which is a metabolic byproduct • In an acidic environment, hemoglobin tends to give up oxygen and bind the protons that are in the tissues • Extra protons are there as the result of the acid CO2 • Bicarbonate is moved out of the RBC, exchanged with Chloride (Cl-).
Gas Exchange • Know that pH and Gas Pressures drive respiration • Know which direction the equation is going H2O + HbO2- + CO2 HHb + O2 + HCO3- Which one occurs in the lungs? Which one occurs in the peripheral tissues?