The Structure of the Atom
950 likes | 1.88k Vues
The Structure of the Atom. Chap. 4. I. History. I. History. A. Early Greeks. Explain matter with 4 ‘elements’. I. History. A. Early Greeks. 1. Air 2. Earth 3. Fire 4. Water. Explain matter with 4 ‘elements’. I. History. A. Early Greeks B. Democritus.

The Structure of the Atom
E N D
Presentation Transcript
The Structure of the Atom Chap. 4
I. History A. Early Greeks Explain matter with 4 ‘elements’
I. History A. Early Greeks 1. Air 2. Earth 3. Fire 4. Water Explain matter with 4 ‘elements’
I. History A. Early Greeks B. Democritus Greek philosopher who first introduced concept of ‘atomos’
I. History A. Early Greeks B. Democritus C. Alchemists Devoted to making precious metals from base metals
I. History A. Early Greeks B. Democritus C. Alchemists D. Dalton Formulated the ‘Atomic Theory of Matter’
Atomic Theory of Matter 1. All matter is made of . . .
Atomic Theory of Matter 1. All matter is made of . . . 2. Atoms cannot be . . .
Atomic Theory of Matter 1. All matter is made of . . . 2. Atoms cannot be . . . 3. Atoms of a given element . . .
Atomic Theory of Matter 1. All matter is made of . . . 2. Atoms cannot be . . . 3. Atoms of a given element . . . 4. Atoms of 2 different elements . .
Atomic Theory of Matter 1. All matter is made of . . . 2. Atoms cannot be . . . 3. Atoms of a given element . . . 4. Atoms of different elements . . . 5. In a chemical reaction atoms are
I. History • A. Early Greeks • B. Democritus • C. Alchemists • Dalton • Definition of Atom
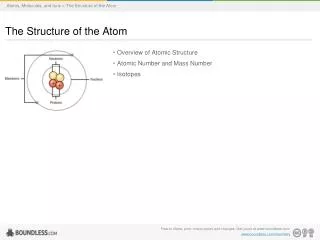
Atom The smallest particle of an element that retains properties of that element.
II. Discoveries of atoms • J.J. Thomson – 1890s Used a cathode ray tube (CRT) to measure the charge:mass ratio of an electron
II. Discoveries of atoms • J.J. Thomson – 1890s • chg:mass = • 1.76 x 1011 C/kg
II. Discoveries of atoms • J.J. Thomson – 1890s • chg:mass = • 1.76 x 1011 C/kg • Discovery led to plum-pudding model
II. Discoveries of atoms • J.J. Thomson – 1890s • Robert Millikan - 1909 Measured the charge of an electron in his ‘oil drop’ exper.
II. Discoveries of atoms • J.J. Thomson – 1890s • Robert Millikan - 1909 • Charge = 1.6 x 10-19
II. Discoveries of atoms • J.J. Thomson – 1890s • Robert Millikan - 1909 • Rutherford - 1911 Discovered nucleus in gold-foil experiment
II. Discoveries of atoms • J.J. Thomson – 1890s • Robert Millikan - 1909 • Rutherford - 1911 • Fired alpha particles at thin metal sheet.
II. Discoveries of atoms • J.J. Thomson – 1890s • Robert Millikan - 1909 • Rutherford - 1911 • Fired alpha particles at thin metal sheet. • Expected them to go straight through, but some deflected.
II. Discoveries of atoms • J.J. Thomson – 1890s • Robert Millikan - 1909 • Rutherford - 1911 • Fired alpha particles at thin metal sheet. • Expected them to go straight through, but some deflected. • This led to nuclear model.
II. Discoveries of atoms • The atom
II. Discoveries of atoms • The atom • The electron was discovered first
II. Discoveries of atoms • The atom • The electron was discovered first • The proton was described by Rutherford
II. Discoveries of atoms • The atom • The electron was discovered first • The proton was described by Rutherford • The neutron was described by Chadwick
III. Representing Atoms What makes a carbon atom different from a nitrogen atom?
III. Representing Atoms • The atomic number
III. Representing Atoms • The atomic number • The number of protons
III. Representing Atoms • The atomic number • The number of protons • Written on the periodic table.
III. Representing Atoms • The atomic number • The number of protons • Written on the periodic table. • This will equal the number of electrons, too.
III. Representing Atoms • The atomic number • The mass number
III. Representing Atoms • The atomic number • The mass number • The number of protons + neutrons
III. Representing Atoms • The atomic number • The mass number • The number of protons + neutrons • Always a whole number
III. Representing Atoms • The atomic number • The mass number • Isotopes
III. Representing Atoms • The atomic number • The mass number • Isotopes • Atoms with the same number of protons, different number of neutrons
III. Representing Atoms • The atomic number • The mass number • Isotopes • Atoms with the same number of protons, different number of neutrons • Isotopes have same properties, but different masses
III. Representing Atoms • The atomic number • The mass number • Isotopes • Notation
Nuclide Symbol Notation Cl 37 17
Nuclide Symbol Notation Element symbol Cl 37 17
Nuclide Symbol Notation Cl 37 17 Atomic number
Nuclide Symbol Notation Mass number Cl 37 17
Self Check – Ex. 1 Write the nuclide symbols for elements with these particles: Nuclide #1 22 protons 24 neutrons Nuclide #2 22 protons 26 neutrons
Self Check – Ex. 2 How many protons, neutrons, and electrons are in this element? 95 Mo 42
Self Check – Ex. 3 How many protons, neutrons, and electrons are in this element? 40 K
IV. Mass of atoms • Measured in amu
IV. Mass of atoms • Measured in amu • Protons and neutrons both weigh about 1 amu (neutrons are a bit more)
IV. Mass of atoms • Measured in amu • Protons and neutrons both weigh about 1 amu (neutrons are a bit more) • The amu is defined as 1/12 the mass of carbon-12