Introduction to the Periodic Table
530 likes | 1.02k Vues
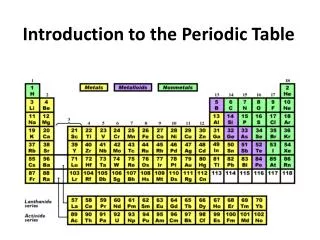
Introduction to the Periodic Table. Atomic Number ● Symbol ● Atomic Weight Element ● Compound ● Mixture. I am Dmitri Mendeleev!. I made the PERIODIC TABLE !. What is the PERIODIC TABLE?. Shows all known elements in the universe. Organizes the elements by chemical properties.

Introduction to the Periodic Table
E N D
Presentation Transcript
Introduction to the Periodic Table Atomic Number ● Symbol ● Atomic Weight Element ● Compound ● Mixture
I am Dmitri Mendeleev! I made the PERIODIC TABLE !
What is the PERIODIC TABLE? • Shows all known elements in the universe. • Organizes the elements by chemical properties.
What is the ATOMIC NUMBER? • The number of protons found in the nucleus of an atom Or • The number of electrons surrounding the nucleus of an atom.
What is the SYMBOL? • An abbreviation of the element name.
What is the ATOMIC WEIGHT? • The number of protons and neutrons in the nucleus of an atom.
Groups/Families The vertical columns of the periodic table are called groups or families. They are numbered from 1 to 18 from left to right. Each group contains elements with similar chemical & physical properties. They will form the same kinds of ions.
Periods The horizontal rows of elements in the periodic table are called periods arranged from 1-7. Elements that are close to each other in the same period tend to be more similar than elements that are farther apart.
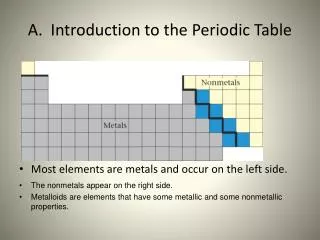
Major Categories: Metals Nonmetals Metalloids- haveproperties that are intermediate between metals and nonmetals.
GROUPS The periodic table is arranged by increasing atomicnumber. (# of protons) Elements in the same group will act the same as other elements in that group.
METALS +1 On the LEFT side of the periodic table +2
METALS 1-12 • Properties of Metals • Shiny • Malleable (bendable) • Good conductor of heat and electricity • Usually solids at room temperature
NON-METALS -1 On the RIGHT side of the periodic table -2
NON-METALS 1-12 17-18 • Properties of Non-Metals • Not shiny (dull) • Brittle (breaks easily) • Poor conductor of heat and electricity • Usually liquids and gases at room temperature • Most reactive non-metals are group 17 (halogens) • Group 18 (noble gases) are non-reactive
METALLOIDS Along the STAIRCASE of the periodic table
METALLOIDS 1-12 7-8 B, Si, Ge, As, Sb, Te, Po, At • Properties of Metalloids • Show properties of metals and non-metals
Group I Alkali Metals • not including hydrogen. • require the least energy to lose one electron. • Turns into a +1 ions • Very reactive metals, always combined with something else in nature (like in salt).
Group 2 Alkaline Earth Metals • Can turn into +2 ions • Reactive metals that are always combined with nonmetals in nature. • Several of these elements are important mineral nutrients (such as Mg and Ca
Group 3-12 Transition Metals • Good conductor of heat • Less reactive harder metals • Includes metals used in jewelry and construction.
Group 14 Carbon Family • Contains elements important to life and computers. • Carbon is the basis for an entire branch of chemistry. • Silicon and Germanium are important semiconductors.
Group 15 Nitrogen Family • Can turn into -3 ions • Nitrogen makes up over ¾ of the atmosphere. • Nitrogen and phosphorus are both important in living things.
Group 16 Oxygen Family • Can turn into -2 ions • Oxygen is necessary for respiration. • Many things that stink, contain sulfur (rotten eggs, garlic, skunks,etc.)
Group 17 Halogens • Can turn into -1 ions • Very reactive, volatile • Always found combined with other element in nature .
Group 18 Noble Gases Unreactive, most resistant to forming compounds Are gases at room temperature Have a full valence shell
require the least energy to lose one electron. Group I Group 18 most resistant to forming compounds
How do I find the number of protons, electrons, and neutrons in an element using the periodic table? • # of PROTONS= ATOMIC NUMBER • # of ELECTRONS= ATOMIC NUMBER • # of NEUTRONS= ATOMIC _ ATOMIC MASS NUMBER
How does an atom become an ion? By gaining or losing an electrons
Ions An atom or groups of atom that have positive or negative charge. Formed by gaining or losing electrons Ions are NEVER formed by gaining or losing protons Cations (+) or anions (-)
Cations Positive charge Formed by the atom losing electrons Metals form cations
Anions Negative charge Formed by atoms gaining electrons Non-metals form anions
How do we know what atoms form which ions? Use the periodic table
Isotope What’s in the neutron?
Isotopes Defined Atoms with the same number of protons butdifferent number of neutrons are called isotopes
Mass numbers All carbons have 6 protons C-12 has 6 neutrons C-13 has 7 neutrons C-14 has 8 neutrons Different isotopes weigh a different amount because of the different amount of neutrons. The isotopes are named by their mass number. Remember proton + neutron = mass
shorthand isotopic notation Mass number for this isotope 64 Cu 29 Atomic number for this element Chemists often write out isotopes using a shorthand notation:
Now you are almost as smart as I am! But not as handsome! Man, I look GOOD!