The Periodic Table
The Periodic Table. Our current society takes for granted all of the hard work, research, chance, and luck that has gone into creating and discovering the materials that are used in the products we utilize every day.

The Periodic Table
E N D
Presentation Transcript
Our current society takes for granted all of the hard work, research, chance, and luck that has gone into creating and discovering the materials that are used in the products we utilize every day. For example, who was the first person to set or find a random black rock (coal) on fire and discover that it provided a good, constant source of heat? Who was the first person to discover that a substance found in some rocks was capable of being the ultimate explosive (uranium)? How’d They Come Up With That? Coal Uranium
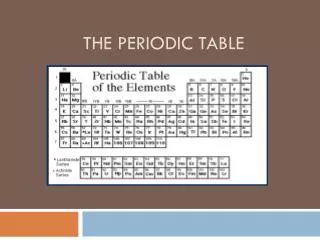
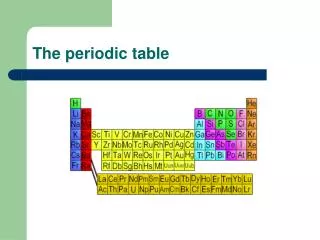
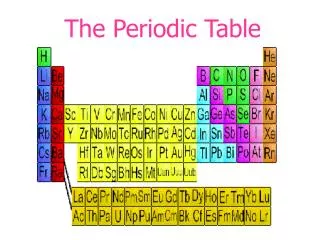
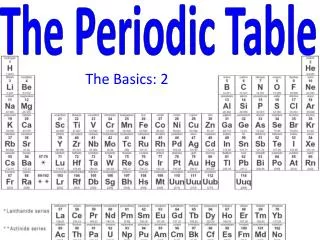
Organizing the elements In nature and in the lab we have discovered over 100 different elements. We’ve organized the elements into a table based on their PHYSICAL and CHEMICAL PROPERTIES It took us almost 2000 years to figure out the properties of the elements currently in the Periodic Table of Elements and arrange them.
I. Developing the Periodic Table • In the early 1800s, enough information was known about the elements that scientists wanted an easy way to categorize the Earth’s ingredients.
So why don’t we just list the elements in a straight line? Why make a table? • An analogy: When you go to the grocery store and are looking for a specific ingredient, you want things to be grouped in a logical way so that you can easily find what you need. • Scientists wanted the elements that they were using in their experiments to also be grouped so that they could easily find elements with the characteristics that they desired.
Developing the Periodic Table • Many methods of organization were tried before scientists found the most effective way of grouping the elements
Dmitri Mendeleev • In 1869 he published a table of the elements organized by increasing atomic mass. 1834-1907
Developing the Periodic Table • Mendeleev stated that the properties of elements are a periodic function of their atomic masses. • If we lay out all of the elements in order by atomic mass, we would see the same properties showing up periodically • For example, as you would begin down the list of the first 18 elements, every 9th (or so) atom would be super reactive and want to gain 1 electron
Lothar Meyer • At the same time, he published his own table of the elements organized by increasing atomic mass. 1830-1895
Both Mendeleev and Meyer arranged the elements in order of increasing atomic mass. • Both left vacant spaces where unknown elements should fit. • So why is Mendeleev called the “Father of the Modern Periodic Table” and not Meyer, or both? Father of the Modern Periodic Table
Mendeleev … • used his periodic function theory and corrected the atomic masses of Be, In, and U. • was so confident in his table that he used it to predict the physical properties of three elements that were yet unknown (Sc, Ga & Ge). • After the discovery of these unknown elements between 1874 and 1885, and the fact that Mendeleev’s predictions were amazingly close to the actual values, his table was generally accepted.
Mendeleev’s Periodic Table • However, in spite of Mendeleev’s great achievement, problems arose when new elements were discovered and more accurate atomic weights determined. • By looking at our modern periodic table, we can identify some problems which might have caused chemists a headache. • Atomic Masses • Ar and K (40-39) • Co and Ni (59-58) • Te and I (128-127) • Th and Pa (232-231)
Henry Moseley • In 1913, through his work with X-rays, he determined the actual nuclear charge (atomic number) of the elements*. He rearranged the elements in order of increasing atomic number. *There is in the atom a fundamental quantity which increases by regular steps as we pass from each element to the next. This quantity can only be the charge on the central positive nucleus. His research was halted when the British government sent him to serve as a foot soldier in WWI. He was killed in the fighting in Gallipoli by a sniper’s bullet, at the age of 28. Because of this loss, the British government later restricted its scientists to noncombatant duties during WWII. 1887-1915
Periodic Law • This arrangement allowed elements with similar properties to be lined up into the same column in the periodic table • Ex. All of the elements in group 1 are extremely reactive when by themselves and want to give away 1 electron
Glenn T. Seaborg • After co-discovering 10 new elements, in 1944 he moved 14 elements out of the main body of the periodic table to their current location below the Lanthanide series. • These became known as the Actinide series. 1912-1999
Glenn T. Seaborg • He is the only person to have an element named after him while still alive. "This is the greatest honor ever bestowed upon me - even better, I think, than winning the Nobel Prize." 1912-1999
Video - Review • Periodic Table History (6:33)
Periodic Table Geography Where are elements found?
The horizontal rows of the periodic table are called PERIODS.
The elements in any group of the periodic table have similar physical and chemical properties! The vertical columns of the periodic table are called GROUPS, or FAMILIES.
General Properties of Metals • Metals are good conductors of heat and electricity • Metals are malleable • Easily bent and shaped • Metals are ductile • Able to be drawn out into a wire • Metals have a shiny luster
Examples of Metals Copper, Cu, is a relatively soft metal, and a very good electrical conductor. Zinc, Zn, is more stable than potassium Mercury, Hg, is the only metal that exists as a liquid at room temperature
General Properties of Metalloids • They have properties of both metals and nonmetals. • Metalloids are more brittle than metals, less brittle than most nonmetallic solids • Metalloids are semiconductors of electricity • Some metalloids possess metallic luster Ex: Silicon possesses a metallic luster, yet it is an inefficient conductor and is brittle.
General Properties of Nonmetals • Nonmetals are poor conductors of heat and electricity • Good insulators • Nonmetals tend to be brittle when solids • Many nonmetals are gases at room temperature Carbon, the graphite in “pencil lead” is a great example of a nonmetallic element.
Examples of Nonmetals Microspheres of phosphorus, P, a reactive nonmetal Sulfur, S, was once known as “brimstone” Graphite is not the only pure form of carbon, C. Diamond is also carbon; the color comes from impurities caught within the crystal structure
The Periodic Table of Elements Write the group names! Chalcogens Halogens P E R I O D Noble Gases Alkali Metals Alkali Earth Metals Transition Metals Transition Metals Transition Metals Transition Metals Lanthanides Actinides
These elements are also called the rare-earth elements. InnerTransition Metals Notice that it continues from period 6 & 7 6 7
Noble Gases Last slide
III. Periodic Law • Before we can discuss the characteristics of each group, we must introduce the Period Law.
Periodic Law • When elements are arranged in order of increasing atomic number, there is a periodic pattern in their physical and chemical properties.
Periodic Table Review • Elements with similar e- configurations are placed in the same group. (same number of e- in last energy level) • The number of electrons in the outermost energy level determines how reactive an element will be • These electrons in the highest energy level are called “Valence Electrons” • Therefore elements in the same group have the same number of valence e-
Group 1 • EX: H = 1s1 Li = 1s22s1 Na = 1s22s22p63s1 • All elements in group 1 have 1 valence electron (1 electron in highest energy level)
Group 17 • Ex: F = 1s22s22p5 Cl = 1s22s22p63s23p5 Br = 1s22s22p63s23p64s23d104p5 • All elements in group 17 have 7 valence electrons (7 electrons in highest energy level)
Valence Electrons 2 1A • Highlight the valence electrons on your periodic table 3A 4A 5A 6A 7A 2A They differ: 2-3 8
Octet Rule • Octet Rule = Eight electrons in an outer level render an atom unreactive (stable). Therefore all atoms are trying to get to this state of 8 valence electrons. • Atoms obtain an octet of electrons in their valence orbital by • taking electrons from another atom, • giving their valence electrons away, • or sharing electrons with another atom.
Octet Rule • Whether an atom gains, loses or shares e- to form an octet depends on how close the atom is to having the 8 e-. • If an atom has less than 4 valence e- then it will try to lose them so that the energy level below (which is full w/ an octet) will then be considered the outermost energy level. • Ex: Na = 1s22s22p63s1 • If Sodium loses 1 electron in the 3s energy level, then energy level 2 will be considered the outermost energy level. Since it has an octet the atom will be stable.
Octet Rule • If an atom has more than 4 valence electrons it will try to steal electrons from other atoms to form an octet • Ex: Cl = 1s22s22p63s23p5 • If Chlorine gains 1 electron it will have an octet and be stable.
Octet Rule • Atoms with 4 electrons in their valence shell will share4 electrons with other atoms to feel as though they have an octet. Last slide
IV. Oxidation Numbers • Our knowledge of e- configurations and the stability of noble gases allows us to predict oxidation numbers for elements. • Oxidation numbers represent the charge an ion obtains after losing or gaining valence electrons. Ex. Ca = 1s22s22p63s23p64s2 2 valence e-, will want to get rid of them so that the full 3rd energy level (8 electrons) will then be it’s outermost shell Ca2+=1s22s22p63s23p6