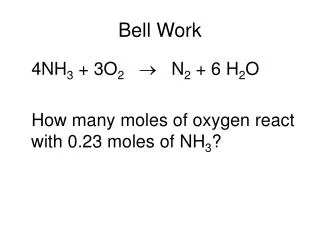Bell Work
Explore the chemical reaction of ammonia (NH3) with oxygen (O2) producing nitrogen (N2) and water (H2O). Learn how to determine the limiting reagent by calculating the moles of oxygen required for 0.23 moles of NH3. Engage with a virtual lab to balance the chemical equation, identify limiting reagents, and perform calculations for CO2 production. Gain insights into the differences between limiting and excess reagents, and discover how concepts change when using mass instead of moles in reactions.

Bell Work
E N D
Presentation Transcript
Bell Work 4NH3 + 3O2 N2 + 6 H2O How many moles of oxygen react with 0.23 moles of NH3?
Open the Link • virtual lab link
C 1 1 H 4 2 O 2 3
ID the limiting reagent. How do you know?
ID the limiting reagent. How do you know?
Balance the equation Go to the virtual lab and check the answer.
Go to the virtual lab and select different number of moles. Copy and fill in the table on the next page. 2 reactants that you can control
Circle the Limiting Reagent, Calculate the moles CO2, Calculate Excess
Questions What would be different if you had mass instead of moles? What is the difference between a limiting reagent and excess reagent?