Coagulation
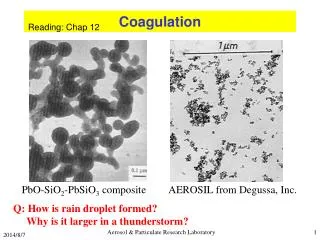
Coagulation. Reading: Chap 12. PbO-SiO 2 -PbSiO 3 composite. AEROSIL from Degussa, Inc. Q: How is rain droplet formed? Why is it larger in a thunderstorm? .

Coagulation
E N D
Presentation Transcript
Coagulation Reading: Chap 12 PbO-SiO2-PbSiO3 composite AEROSIL from Degussa, Inc. Q: How is rain droplet formed? Why is it larger in a thunderstorm? Aerosol & Particulate Research Laboratory
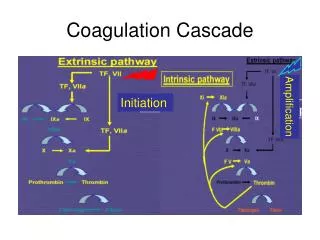
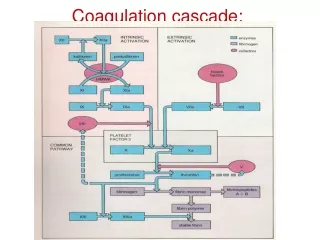
Definition: The process whereby aerosol particles collide with one another due to a relative motion between them and adhere to from larger particles • Thermal Coagulation: due to Brownian motion • Kinematic Coagulation: due to external forces Q: What are the possible external forces? Q: Any difference if solid or liquid particles coagulate? Agglomeration/Aggregation (if particles do not merge) Coalescence (sintering) Q: Do solid particles stay agglomerated? Q: What are the effects of coagulation on aerosol size distribution? Aerosol & Particulate Research Laboratory
Collision Frequency Function(Population Balance) Rate of formation of size k particle Collision Frequency ni: number concentration of size i particle Q: What factors affect the frequency? Rate of removal of size k particle Net rate of size k particle Aerosol & Particulate Research Laboratory
Brownian Coagulation Continuum regime b1,2 Free molecular regime dp1 Q: Any trend? What’s the physics behind it? Aerosol & Particulate Research Laboratory
Nearly Monodisperse System Collision frequency function Coagulation equation Summing over all values of k Integrating once Aerosol & Particulate Research Laboratory
Q: How do you know if coagulation is the main mechanism in your experimental system? Aerosol & Particulate Research Laboratory
Q: How long does it take for N = 1012 #/cc to reduce to its half conc.? N = 106? Q: How to slow down coagulation? Q: If I have the final concentration and particle size, can I determine the original PSD if coagulation is the major mechanism? Aerosol & Particulate Research Laboratory
Side note: Aging atmospheric aerosol also approaches asymptotic behavior: become more oxidized, less volatile and more hygroscopic Jimenez et al., Science, 2009, 326, 1525 Aerosol & Particulate Research Laboratory
Coagulation of a Log-Normally Distributed Aerosol (t/coag) Q: How does N change wrt time? Q: What’s the impact of s0 on N? Aerosol & Particulate Research Laboratory
Q: How does sg change wrt time? 2.5 0 = 2.5 2.0 2.0 g1.5 1.5 1.0 1.0 Q: Why does PM2.5 in Shanghai decrease when there is sand storm from Gobi desert? Aerosol & Particulate Research Laboratory
Gas-To-Particle Conversion Reading: Chap 13.5 Q: What’s the interaction between gas molecules and particles? Activated Carbon VOC Water droplet Water vapor Q: Examples? ==>Condensation Q: How do we have the first particle for vapor condensation? ==>Nucleation Q: Examples? Aerosol & Particulate Research Laboratory
(Saturation) Vapor Pressure Time to reach equilibrium How does vapor pressure change if the temperature increases? Vapor Pressure • The pressure required to maintain a vapor in equilibrium with the condensed vapor (liquid or solid) with a flat surface at a specified temperature Partial Pressure) Pv in mmHg and T in oC(if Table 9.2 is used) For water, A = 8.10765. B = 1,750.286, C = 235 Aerosol & Particulate Research Laboratory
Saturation Ratio (or relative humidity for water) Supersaturation: S > 1 (RH > 100%) Q: What would happen if S > 1? Q: When does it happen for S > 1? Adiabatic Expansion, Mixing, Cooling, Speciation change by Gas Phase Reaction Aerosol & Particulate Research Laboratory
Adiabatic Expansion An expansion allowing no heat input from the surroundings Q: Saturated water vapor @ 20 oC (p = 17.6 mmHg) expands 20%. Calculate the new saturation ratio. Ps @ 0 oC = 4.7 mmHg, k = 1.38. Example: Cloud formation, smoke generated in the neck of a wine bottle Aerosol & Particulate Research Laboratory
Kelvin Effect Change in Gibbs free energy accompanying the formation of a single droplet of pure material A of radius Rp (initial) (final) NT: # of molecules Nu: # of vapor molecules Nl: # of liquid molecules gu: Gibbs free energy of a molecule in the vapor phase gl: Gibbs free energy of a molecule in the liquid phase ul: volume of one liquid molecule s: surface tension Rp: particle radius A system moves toward a lower energy state whenever possible. Aerosol & Particulate Research Laboratory
Q: How does DG change as Rp increases? Aerosol & Particulate Research Laboratory
Maximizing the G (Critical radius) (Critical Number) (Max G) (Kelvin Equation) (Kevin Ratio) The partial pressure of vapor at the surface of a small droplet is greater than the saturation vapor pressure defined for a flat surface Aerosol & Particulate Research Laboratory
Critical number and Radius for water droplets Equilibrium vapor pressure over a pure water droplets for various dp at T = 298 K Aerosol & Particulate Research Laboratory
Condensation Reading: Chap 13 • In the continuum regime, diffusion theory is used. At steady state Rate of diffusional condensation Volume growth Size growth Aerosol & Particulate Research Laboratory
Q: How does the growth rate depend on particle volume? Particle size? Implication? Ex: For dp = 0. 1 and 1 mm, calculate the new dp after 1 sec of water vapor condensation at 20 oC. • In the free molecular regime, the gas kinetic theory is applied Volume growth Size growth Q: How can the condensation rate be increased? Q: How does the growth rate depend on size? Implication? Aerosol & Particulate Research Laboratory
Condensation Nuclei Counter Q: How to measure N by light scattering intensity if dp is different? Q: Can we use CNC to measure dp? Q: What if we combine a DMA and a CNC? Aerosol & Particulate Research Laboratory
Condensation with Soluble Nuclei & Ions Saturation ratio for droplets containing dissolved materials m: mass of the dissolved salt i: number of ions each molecule of salt forms Mw: molecular weight of solvent (water) Ms: molecular weight of salt : density of solvent Aerosol & Particulate Research Laboratory
Saturation ratio vs droplet size for 10-16 g NaCl (Solid sphere of 0.045 mm in diameter) Aerosol & Particulate Research Laboratory
Relative humidify vs droplet size (a larger scale) Droplet formation & crystalization 10-14g Q: Implication? Aerosol & Particulate Research Laboratory
Evaporation • The reverse process of particle growth though there is no ciritical size. • Droplet lifetime/drying time Q: Where is droplet lifetime important? • For volatile materials, cooling of the droplet due to the latenet heat of evaporation needs to be considered. D = 0.24 cm2/s for water vapor R = 62400 mmHg·cm3/K·mol Droplet lifetime for dp > 1.0 mm Aerosol & Particulate Research Laboratory
Q: For a given aerosol system, what’s the effect of heating followed by cooling? Condensation Aerosol Generator Aerosol & Particulate Research Laboratory
Summary Aerosol & Particulate Research Laboratory
D = 0.24 cm2/s 1 = 3×10-23 cm3 k = 1.38×10-16dyne·cm/K T = 298 K p1 ̶ pd = 1.01×106 dyne/cm2 t = 1 s Aerosol & Particulate Research Laboratory