Understanding Chemical Bonding: Types, Characteristics, and Examples
This text explores the concept of chemical bonding, focusing on how atoms stabilize by achieving full outer electron shells, transitioning into compounds that exhibit distinct properties from their constituent elements. It outlines three main types of bonding: ionic (electron transfer), covalent (electron sharing), and metallic (free electron movement). Examples such as sodium chloride highlight the transformation from individual elements to stable compounds. The nature of ions—whether positive or negative—is also discussed, emphasizing their role in the bonding process.

Understanding Chemical Bonding: Types, Characteristics, and Examples
E N D
Presentation Transcript
• Used to stabilize an atom = Atoms want to have a full outermost electron shell / energy level. • Turning atoms into Happy atoms
• Atoms are CHEMICALLY bonded. *(This is the definition of a Compound) = Is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms *** Properties of the new substance (the compound) are different from the original elements.
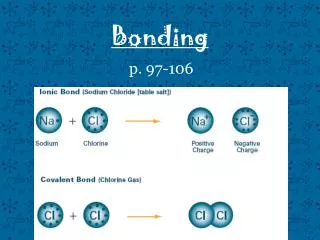
• Some examples: Sodium Soft, silvery white metal. Reacts violently with water. Chlorine Poisonous Greenish – Yellow gas NaCl = Sodium Chloride. Salt (Yummy on French Fries.)
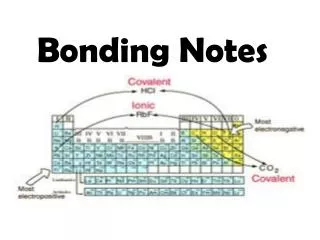
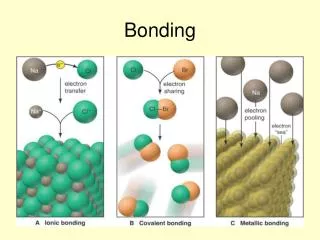
• Three main types of bonding: 1. Ionic bonds is transfering electrons. ** Occurs between a metal and a non metal
Covalent bonds is the sharing of electrons. ** Occurs between two non metals
3. Metallic – Have free electrons from outer shells ** Occurs between two metals • Metal atoms form positve ions and act like buoys (floaty pool toys) and lifeguards for the negative electrons to swim around / be attracted to.
Ions: What makes an atom an ion? Having a different number of protons and electrons • Can be positive or negative • Results from atoms gaining or losing electrons. * Negative ions have gained an extra e- (now they’re extra negative and have more e- than p+) * Positive ions have lost an e- (now they’re a less negative = positive and they have more p+ than e-)