Amino acids and proteins
150 likes | 326 Vues
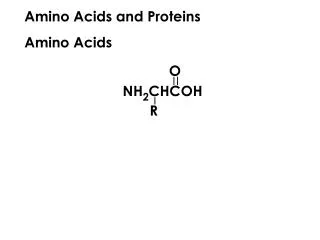
Amino acids and proteins. Precipitation of Proteins at isoelectric Point. Protein solubility: The solubility of proteins in aqueous buffers depends on the distribution of hydrophilic and hydrophobic amino acid residues on the protein’s surface.

Amino acids and proteins
E N D
Presentation Transcript
Precipitation of Proteins at isoelectric Point • Protein solubility: • The solubility of proteins in aqueous buffers depends on the distribution of hydrophilic and hydrophobic amino acid residues on the protein’s surface. • Proteins that have high hydrophobic amino acid content on the surface have low solubility in an aqueous solvent. • Charged and polar surface residues interact with ionic groups in the solvent and increase solubility.
B. Isoelectric point precipitation • Isoelectric point (pI):is the pH-value of a solution at which the total net charge of a protein equals zero. • At a solution pH that is above the pI the surface of the protein is predominantly negatively charged and therefore like-charged molecules will exhibit repulsive forces. • Likewise the surface of the protein is predominantly positively charged at a solution pH that is below the pI, and repulsion between proteins occurs. • However, at the pI the negative and positive charges are eliminated, repulsive electrostatic forces are reduced and the dispersive forces predominate.
The dispersive forces will cause aggregation and precipitation. • The pI of most proteins ranges between the pH 4 to 6. • When microorganisms grow in milk, they often produce acids and lower the pH of the milk. • The phenomenon of precipitation or coagulation of milk protein (casein) at low pH as milk becomes spoiled is one of the common examples of protein isolation due to changes in the pH.
Procedure • Into a 50 ml volumetric flask add 20 ml of water. • Add 0.25 g of pure casein, followed by the addition of 5 ml of 1 N NaOH solution. • Once casein is dissolved, add 5 ml of 1 N acetic acid solution, then dilute with H2O to 50 ml and mix well. The resulted solution is a 0.1 N casein acetate sodium.
Setup a series of 9 test tubes. • In the first test tube put 3.2 ml1 N CH3COOH, and 6.8 ml H2O and mix thoroughly. • In each of the other test tubes (2-9) put 5 ml H2Od. • From the test tube 1 transfer 5 ml to the test tube 2, and mix thoroughly. • Repeat step 7 for the rest of test tubes (3 - 9).
Now to each test tube (1 -9) add 1 ml of the casein acetate sodium solution, and shake the test tubes immediately. • Let the samples stand for 30 min, and note the turbidity in the 9 test tubes. • Use) +( and )– (signs to describe the turbidity in the different test tubes. • You should observe the most precipitation in the test tube which has the pH around 4.7 (close to the isoelectric point of casein).
Isolation of Casein from Milk • Milk is a food of exceptional interest not only is milk an excellent food for the very young, but humans have also adapted milk, specifically cow’s milk, as a food substance for persons of all ages. • Many specialized milk products like cheese, butter, and ice cream are staples of our diet. • Milk is probably the most nutritionally-complete food that can be found in nature. This property is important for milk, since it is the only food young mammals consume in the nutritionally significant weeks following birth.
Whole milk contains vitamins (Thiamine, Riboflavin, and vitamins A, D, and K), minerals (calcium, potassium, sodium, phosphorus, and trace metals), proteins (which include all the essential amino acids), carbohydrates (chiefly lactose), and lipids. • The only important elements in which milk is seriously deficient are iron and Vitamin C. • Infants are usually born with a storage supply of iron large enough to meet their needs for several weeks. Vitamin C is easily secured through an orange juice supplement. • There are three kinds of proteins in milk: caseins, lactalbumins, and lactoglobulins. All are globular.
Casein • Is a phosphoprotein, which has phosphate groups attached to some of the amino acid side chains. • casein is a mixture of at least three similar proteins, which differ primarily in molecular weight and amount of phosphorus they contain (number of phosphate groups). • Casein exists in milk as the calcium salt, calcium caseinate, this salt has a complex structure. • It is composed of α, β, and κ caseins which form a micelle, or a solubilized unit.
Neither the α nor the β casein is soluble in milk, singly or in combination. • If κ casein is added to either one, or to a combination of the two, however, the result is a casein complex that is soluble owing to the formation of the micelle. • A structure proposed for the casein micelle is shown in the following figure. The κ casein is thought to stabilize the micelle. Since both α and β casein are phosphoproteins, they are precipitated by calcium ions.
Procedure • Place 20 ml (20 g) of milk into a 125 ml flask and heat at 40 oC in a water bath. • Add 5 drops of glacial acetic acid and stir for about 1 min. • Filter the resulting mixture through 4 layers of cheesecloth held in a funnel and gently squeeze out most of liquid. • Remove the solid (casein and fat) from the cheesecloth, place it into a 100 ml beaker and add 10 ml of 95% ethanol. • Stir well to break up the product. Pour off the liquid and add 10 ml of 1:1 ether-ethanol mixture to the solid. • Stir well and filter through 4 layers of cheesecloth.
Let the solid drain well, then scrape it into a weighed filter paper and let it dry in the air. • Calculate the casein percentage in milk as follows: % Casein = (grams of casein \ grams of milk) x 100