Amino acids and proteins
Amino acids and proteins. Dr Una Fairbrother. Amino acids and proteins. Proteins are composed of amino acids When a protein is hydrolysed e.g. with hydrochloric acid then about 20 different amino acids can be detected

Amino acids and proteins
E N D
Presentation Transcript
Amino acids and proteins Dr Una Fairbrother
Amino acids and proteins • Proteins are composed of amino acids • When a protein is hydrolysed e.g. with hydrochloric acid then about 20 different amino acids can be detected • When a protein is sequenced it may be found that it is composed of hundred amino acids • Therefore each of the different amino acids is occurring more than once
Each protein has unique sequence • Think of coloured beads in a necklace • One necklace may differ from another by the order of the beads and the frequency with which they occur • Different amino acid content and sequence alters the properties of proteins
Amino acid game • http://www.wiley.com/legacy/college/boyer/0470003790/animations/animations.htm
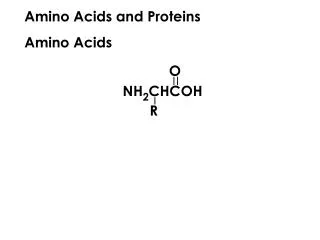
General Structure of an alpha amino acid • A 'free' amino acid (a single amino acid) always has: • An amino group -NH2, • A carboxyl group -COOH • A hydrogen -H • A chemical group or side chain -"R". • These are all joined to a central carbon atom, the a -carbon
Only L Amino Acids Are Found in Proteins. • The L and D Isomers of Amino Acids. • R refers to the side chain. • The L and D isomers are mirror images of each other.
R groups • Differences between amino acids due to differing R group structures • The R group or side chain can be aliphatic, hydroxyl or sulphur containing, aromatic, basic or acidic
Features of the R group • The aliphatic amino acids have hydrocarbon side chains which are hydrophobic. • They do not like to be in contact with water molecules in an aqueous solution. • These allow hydrophobic bonding in proteins to occur. • For this reason, they are often located in the core of the protein, surrounded by the rest of the protein, and "shielded" by them from the aqueous surroundings.
Properties of the aliphatics • Glycine (Gly; G) • simplest and smallest of all, has a single hydrogen as it's side chai • Alanine (Ala; A) • has a methyl group as it's sidechain. • Valine (Val; V) • Longer sidechain with a branch. As the aliphatic side chains get longer they are also more hydrophobic. • Leucine (Leu; L) • has another methyl group attached to the sidechain. • Isoleucine (Ile; I) • similar to L and V but orientation of sidechain atoms is different. Isoleucine also has two centers of asymmetry • Proline (Pro; P) • Different- the sidechain is bonded to the a-carbon and the amino group. Marked effects on the architecture of proteins. Less hydrophobic than the others.
Proline • Proline is a special case • Due to its structure it causes bends in proteins • It is a feature which contributes to the compactness of globular proteins
Neutral Polar Amino Acids • These amino acids are not charged at physiological pH. • Serine and threonine, asparagine and glutamine have polar hydroxyl groups in their side chains • These can contribute to hydrogen bonding in proteins • For this reason the amino acids are classed as hydrophillic.
Sulphur containing amino acids • Cysteine (Cys; C) • contains a thiol or sulphydryl group (-SH). • This is extremely reactive, and can form hydrogen bonds. • But the long aliphatic part of the side chain makes it quite hydrophobic. • The (-SH) group of cysteine can be oxidised with another cystein -SH to give a -S-S-disulphide bridge • Methionine (Met; M) • is a very special amino acid in that it is the "start" amino acid in the process of translation (protein synthesis). • sulphur atom in a thioether linkage, and is relatively unreactive. • Has a highly hydrophobic sidechain.
Aromatic amino acids • Two of the aromatic amino acids, tryptophan and tyrosine are useful in quantitative protein determination. • They are detected in the Lowry assay: a biochemical test to measure the amount of protein in a sample. • Copper(II) ion in alkaline solution reacts with protein to form complexes, which react with a Folin-phenol reagent. The product becomes reduced and can be detected colourimetrically by absorbance at 650 nm.
Aromatic amino acids • Contain an aromatic ring as part of their sidechains. • Because of the aromatic rings, they are highly hydrophobic • Phenylalanine (Phe; F) • Contains a phenyl ring attached to a methylene group • Tyrosine (Tyr; Y) • contains a hydroxyl group on the phenyl ring • makes it less hydrophobic than F. It is also a reactive group • Tryptophan (Trp; W) • has a slightly different ring attached to the methylene group. • This is an indole ring and it is highly hydrophobic.
Charge • The basic and acidic amino acids can exist in charged forms either -ve (with acidic aas) or +ve (with basic aas). • Attraction between oppositely charged amino acids in proteins produces ionic bonds.
Acidic Amino Acids • These amino acids are highly polar, and are nearly always negatively charged at physiological pH. • Aspartate (Asp; D) • is really aspartic acid. • It is called aspartate because it is usually negatively charged at physiological pH and so it is named for the carboxylate anion. • Glutamate (Glu; E) • is also called glutamic acid. • The side chain of glutamate also has a carboxylate group which has a negative charge at physiological pH
Basic Amino Acids • Contain side chains which are postively charged at physiological pH. • Lysine (Lys; K) • one of the longest side chains • it is very polar because of the terminal amino group and is a hydrophillic amino acid. • Arginine (Arg; R) • the largest of all sidechains • Bacause of the guanidino group on the sidechain it is postively charged at physiological pH. • Histidine (His; H) • has an imidazole ring which often sits inside the active site of an enzyme and helps bonds to be broken or made. • It can do this because it can exist in two states -uncharged, or positively charged.
Ion Pairs • When amino acid sidechains of opposite charge are in close proximity, they can form an ion pair (also called a salt bridge). • If they can form a salt bridge, they will usually be buried. • Since charge is affected by pH, so is the formation and the breakage of these ion pairs. • Salt bridges increase the stability of the tertiary structure.
Features of the a-amino and a-carboxyl group • In solution amino acids can exist in different charged forms depending on pH. • At the most acid pH values they are fully protonated with two weakly acidic groups.
Dissociation constant (pKa) values • If we know the pKa value for a protein titratable group, we can predict the charge present on this group if the protein is in solution at a given pH value, since pH=-log([H3O+]) • Glycine contains two ionisable groups: an a-carboxyl group and a protonated a-amino group. • As base is added, these two groups are titrated. • The pKa of the a-COOH group is 2.4, whereas that of the a-NH3+ group is 9.8. • The pKa values of these groups in other amino acids are similar. • Some aas, such as aspartic acid, have an ionisable side chain. • The pKa values of ionizable side chains in amino acids range from 3.9 (aspartic acid) to 12.5 (arginine).
Zwitterion • At a neutral pH, both the amino and the carboxyl groups are ionised • giving what is termed the zwitterionic form of the molecule (or dipolar ion).
Titration of the a-Carboxyl and a-Amino Groups of an Amino Acid. • First the carboxyl group then the protonated amino group loses its proton as the pH increases.
Ionisation State as a Function of pH: the graph • The ionisation state of amino acids is altered by a change in pH. • The zwitterionic form predominates near physiological pH.
Titration with NaOH • The curve produced is really two curves added together. On the left-hand side is the curve from the titration of the carboxyl group. On the right-hand side is the curve from the titration of the amino group. • One equivalent of NaOH is required to titrate each acidic group. • When the first equivalent has been added the amino acid is in its dipolar form.
Question • What are the relative amounts of structure I and structure II when 0.5 equivalents have been added? • There are equal amounts of each
Hydrogen Bonds • When unfolded, all polar/hydrophillic sidechains can interact via H-bonds with water. • When the protein folds, they must H-bond to each other and exclude much of the water. • All groups capable of forming a hydrogen bond MUST, hence H-bonding in the backbone (C=O to N-H) by way of helices and sheets is an efficient way of ensuring maximum H-bonding. • Sidechains can either accept (as in C=O) or donate (as in N-H, or O-H) an H-bond. • The capacity of proteins to form hydrogen bonds is an important determinant of protein stability. • Hydrogen bonds can be: • between backbone groups, as in helices and sheets; • between side chains, such as serine or threonine O-H groups and carbonyl carbons of side chains (-C=O); • and between backbone groups and side chain groups.
Summary • General features • Aliphatic amino acids • Neutral polar amino acids • Sulphur containing amino acids • Aromatic amino acids • Charge: • Acidic amino acids • Basic amino acids • Ion pairs • pKa values • Titration with NaOH • Hydrogen bonds • Question
Reading • Biochemistry, 5th edition, Berg, Tymoczko and Stryer • http://www.wiley.com/legacy/college/boyer/0470003790/animations/animations.htm