CYTOSKELETON (I) Actin filaments
712 likes | 2.18k Vues
CYTOSKELETON (I) Actin filaments. Cell Biology Lecture 9. Readings and Objectives. Reading Russell : Chapter 1 (not complete information) Cooper: Chapter 12 Objectives Actin and actin filament dynamism Actin bundles and network Actin and myosin: role in contractile assemblies

CYTOSKELETON (I) Actin filaments
E N D
Presentation Transcript
CYTOSKELETON (I)Actin filaments Cell Biology Lecture 9
Readings and Objectives • Reading • Russell : Chapter 1 (not complete information) • Cooper: Chapter 12 • Objectives • Actin and actin filament dynamism • Actin bundles and network • Actin and myosin: role in contractile assemblies • Role in cellular movement
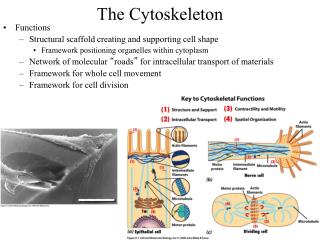
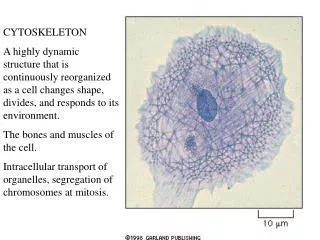
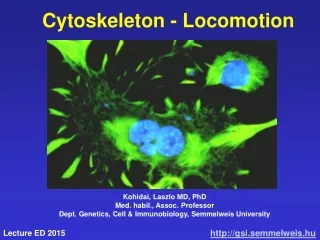
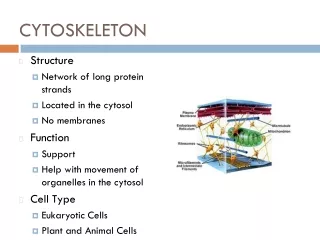
Introduction: Cytoskeleton • A network of protein filaments and tubules, extends from the nucleus to the plasma membrane • Structural framework • cell shape, localize organelles, general organization of the cytoplasm • Movement • Cell movement, internal transport of organelles, muscle contraction • Dynamic structures, continually reorganized Composed of three main types of protein filaments: • Actin filaments (7 nm) • Intermediate filaments (8-11 nm) • Microtubules ( 25 nm)
Structure and Organization of Actin Filaments • Actin filaments (microfilaments): Polymer of Actin, flexible fibers 7 nm in diameter, several µm in length • Actin first isolated from muscle cells in 1942 • Abundant in all types of eukaryotic cells • Mammals have 6 actin genes: 4 are expressed in muscle cells and 2 in nonmuscle cells • Highly conserved • Prokaryotic ancestor is MreB
Actin and Actin Filament • 3-D structure determined in 1990 • Actin: globular (G-actin), 375 aa (43 kD), barbed and pointed ends, binds head-tail to nucleate a trimer • Filamentous (F-actin): monomers added to both end • Filament is polar pointed end vs barbed end
G-Actin and Actin Filament • Polymerization is reversible • The rate at which monomers are added to filaments is proportional to their concentration • ATP bound actin binds to barbed end with high affinity • ADP-actin has low affinity to the pointed ends • when ATP hydrolyses to ADP • ADP-actin dissociates from filaments more readily than ATP-actin • Therefore, the critical concentration of actin monomers is higher for addition to the pointed end than to the barbed end of actin filaments
Treadmilling: polarity of F-actin growth • At cellular actin concentrations • Barbed end of a filament grows 5–10 times faster than the pointed end • ADP-actin dissociates from pointed end • Exchange of ATP for ADP added to barbed-end • Process is calledTreadmilling • Dynamic growth • Direction?PointedBarbed
Actin Binding Proteins • Actin Binding Proteins (ABP): modulate the Assembly and disassembly of actin filaments • ABD/Actin interaction has diverse functionality • Contribute to the cellular role of actin filaments
Actin Remodeling • Some actin-binding proteins bind along the length of actin filaments, stabilizing them or cross-linking them to one another • Others stablize by capping the ends and preventing dissociation • Others promote dissociation, while others regulate the exchange of ATP for ADP.
Initiation of Actin Filament polymerization • Nucleation is the rate-limiting step • Formin and the Arp2/3 complex determine where filaments are formed by facilitating nucleation • Formins nucleate long unbranched actin filaments
Initiation of Actin Filament polymerization • actin filaments actively turn over and branch extensively • These filaments are nucleated by the Arp2/3 (Actin Related Protein) complex, which binds actin/ATP near the barbed ends
Actin Filament Depolymerization • The ADF/cofilin (Actin Depolymerizing Factor) family modifies existing filaments • enhance the rate of dissociation of actin/ADP monomers from the pointed end, and remain bound to the monomers, preventing their reincorporation
Actin Filament Severing • ADF/cofilin can also bind to and sever actin filaments • Profilin reverses the ADF/Cofilin effect • Stimulate exchange of bound ADP for ATP and dissociating the actin/ATP monomers from cofilin • Become available for reassembly in two filaments
Actin filament higher order assemblies • Actin bundles—cross-linked into closely packed parallel arrays • Actin networks—cross-linked in arrays that form 3-D meshworks
Actin Bundles • Parallel filaments cross linked by actin-bundling proteins • Have two domains to bind actin and align the filaments Two types of actin bundles: • Non contractile • filaments (14 nm apart) aligned in parallel, same polarity, barbed ends adjacent to the plasma membrane • Fimbrin: a 68 kD proteins, cross links by its two actin binding domains (ABD)
Actin bundles: IntenstinalMicrovilli • Actin bundles take part in avariety of cell surface protrusions • cell movement • phagocytosis • absorption of nutrients • Intenstinal microvilli: • Membrane projections, increase absorption surface
Actin bundles: IntenstinalMicrovilli • closely packed parallel bundles of 20 to 30 actin filaments • Relatively permanent • The filaments are cross-linked in part by fimbrin and villlin • The actin bundles are attached to the plasma membrane by the calcium-binding protein calmodulin in association with myosin I • At the base attach to actin cortex
Actin bundles: membrane protrusions • Other surface protrusions are transient and form in response to environmental stimuli • Pseudopodia- responsible for phagocytosis and the movement of amoebas • Lamellipodia- broad, sheetlike extensions at the leading edge of fibroblasts • Filopodia- thin projections of the plasma membrane in migrating cells
Actin Bundles: Contractile • Contractile bundles: more widely-spaced filaments (40 nm), cross-linked by α-actinin • α-actinin:a 102 kD protein with single ABD and an α-helical spacer • Interacts with actin as a dimer • Increased spacing allows actin interaction with motor protein myosin II • Important in muscle fiber contraction
Higher order Actin assembly: Actin Network • Filamin (280 kD) form flexible cross-links • Filamin dimer: flexible V-shaped molecule • actin-binding domains at the end of each arm • dimerization domain • Β-sheet spacer • Binds actin orthogonally, form 3-D network beneath the plasma membrane • network (cell cortex) determines cell shape, and cell movement
Actin Network and cell cortex • Red blood cells as mode • lack other cytoskeletal components, so the cortical cytoskeleton is the principal determinant of cell shape • Spectrin, major actin-binding cortex protein • tetramer of two polypeptide chains, αand β • ends of the spectrin tetramers bind actin filaments, resulting in the spectrin-actin network
Actin Network and Cell Cortex • Ankyrin links the spectrin-actin network and the plasma membrane • Protein 4.1 is another link that binds spectrin-actin junctions and the transmembrane protein glycophorin
Association of Actin filaments with Motor proteins • Brings higher level of functional complexity to cells • Cellular or organismal movement • Intracellular cargo transportation, cell division Association with motor protein myosin • Myosin is a molecular motor: converts chemical energy (ATP) to mechanical energy force and movement. • Muscle contraction: model for understanding actin-myosin interactions and the motor activity of myosin molecules
Actin-Myosin and Muscle Contraction • muscle fibers, large cells (50 µm in diameter and up to several centimeters in length) • Cytoplasm consists of myofibrilsmyosin filaments and thin actin filaments • sarcomeres, myofibril units of skeletal and cardiac muscle • actin filaments attached at their barbed ends to the Z disc
Sacromere: a structural and contractile unit • Titin is extremely large protein; extend from the M line to the Z disc • keep myosin II filaments centered in the sarcomere • maintain the resting tension that allows a muscle to snap back if overextended • Nebulin, associated with actin, regulate assembly of actin filaments
Sliding Filament Model • was proposed in 1954 • Myosin slides on actin filament • Sarcomere shortens, bringing the Z discs closer • There is no change in the width of the A band, but the I bands and H zone almost disappear
Sliding Filament Model • Tropomyosin binds along actin filaments, also bound to troponin • No Ca2+, tropomyosin-troponin block binding of myosin to actin • nerve impulses, stimulate release of Ca2+ from the sarcoplasmic reticulum • Ca2+ binds troponin C, shifts the complex • Allows myosin binding to actin
Sliding Filament Model • Myosin II (the type in muscle), large protein with two heavy chains and two pairs of light chains • heavy chains have a globular head region and a long α-helical tail • Tails twist around in a coiled-coil • globular heads bind actin • myosin moves the head groups along the actin filament in the direction of the barbed end
Sliding Filament Model • ATP hydrolysis is required • Binding of ATP dissociates myosin from actin • ATP hydrolysis induces a conformational change that displaces the myosin head group
Sliding Filament Model • myosin head binds to a new position on the actin filament and Pi is released • The “power stroke”: Myosin head returns to its original conformation, which drives actin filament sliding, and ADP is released
Actin and myosin in cell divison • Cytokinesis—division of a cell following mitosis • A contractile ring of actin and myosin II is assembled underneath the plasma membrane • Contraction of the ring pinches the cell in two
Actin and myosin: vesicular transport • Myosin I: much smaller than myosin II,contains a globular head group, acts as a molecular motor • Short tails bind to other structures • Movement of myosin I along actin filament • transport cargo, such as a vesicle