Diffusion Mass Transfer
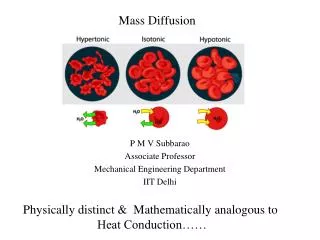
Diffusion Mass Transfer. Mass transfer. Mass transfer is a transport of components under a chemical potential gradient. The component moves to the direction of reducing concentration gradient. The transport occurs from a region of higher concentration to lower concentration.

Diffusion Mass Transfer
E N D
Presentation Transcript
Mass transfer • Mass transfer is a transport of components under a chemical potential gradient. • The component moves to the direction of reducing concentration gradient. Thetransport occurs from a region of higher concentration to lower concentration. • Equilibrium is reached when the gradient is zero. The transport or migration of oneconstituent from a region of higher concentration to that of a lower concentration isknown as mass transfer. Mass transfer operations depend on molecules diffusingfrom one distinct phase to another and are based upon differences in thephysicochemicalproperties of the molecules, such as vapour pressure or solubility. Forinterphase mass transfer, there is a concentration gradient between bulk andinterface., however under steady state, at interface equilibrium is assumed.
Mass transfer operations • Masstransfer operation plays an important role in many industrial processes. • A group ofoperations for separating the components of mixtures is based on the transfer ofmaterial from one homogeneous phase to another. These methods is covered bythe term mass transfer operations which include techniques like: • gas absorptionand stripping, • liquid-liquid extraction, • leaching, distillation, • humidification, • drying, • crystallization and number of other separation techniques.
General Considerations General Considerations • Mass transfer refers to mass in transit due to a species concentration gradient • in a mixture. • Must have a mixture of two or more species for mass transfer to occur. • The species concentration gradient is the driving potential for transfer. • Mass transfer by diffusion is analogous to heat transfer by conduction. • Physical Origins of Diffusion: • Transfer is due to random molecular motion. • Consider two species A and B at the same T and p, but initially separated by a partition. • Diffusion in the direction of decreasing concentration dictates net transport of A molecules to the right and B molecules to the left. • In time, uniform concentrations of A and B are achieved.
Definitions Molarconcentration of species i. Mass density (kg/m3) of species i. Molecularweight (kg/kmol) of species i. Molarflux of species i due to diffusion. Absolute molar flux of species i. Mass flux of species i due to diffusion. Absolute mass flux of species i. ¢¢ : n i Mole fraction of species i Mass fraction of species i Definitions • Transport of i relative to molar average velocity (v*) of mixture. • Transport of i relative to a fixed reference frame. • Transport of i relative to mass-average velocity (v) of mixture. • Transport of i relative to a fixed reference frame.
Property Relations Property Relations • Mixture Concentration: • Mixture Density: • Mixture of Ideal Gases:
L phase is the stream with higher densityV is the one with lower densityExcept for extraction where L is the raffinate and V is the extract
Absorption and stripping- Absorption refers to an operation in which the transfer of material is from a gas phase to a liquid phase. A gas is absorbed by means of liquid in which the solute gas is more or less soluble from its mixture withan inert gas as well as more or less insoluble gas. The liquid is essentiallyimmiscible in the gas phase. The stripping is the separation of gas solute from theliquid phase. The separation of ammonia from an air-ammonia mixture by meansof water is a typical example of absorption. The solute is then recovered from thesolution by distillation. Depending upon situation, both the fluids (absorbent andabsorbing fluid) are reprocessed and/or reused for the operation. Applications of absorption: 1- Hydrogen sulfide(H2S) is removed from hydrocarbon gases by washing with alkaline solution (Amines).2- Washing ethanol vapors from carbon dioxide from molasses fermentor tanks with water to remove ethanol.3- Acetone can be recovered from acetone-air mixture by passing the gas stream into water in which acetone is dissolved while air is passed out. 4-Carbon dioxide present in air is absorbed by sodium hydroxide (NaOH solution) in which chemical absorption takes place. 5- Nitrogen oxides are absorbed in water to give nitric acid. 6- Removal of ammonia coming from coke ovens by water
Adsorption and desorption –It exploits the ability of transfer of mass from • either a gas or a liquid to the surface of a solid. The adsorption is not a true interphasemass transfer operation because the fluid adheres to the solid surface • instead of dissolving in the solid. A desorption involves the transfer of mass from • the solid surface (adsorbents) to the gas or liquid medium (adsorbates). • A fewoperations of practical applications are: • elimination of toxic gases anddeodorization of air • recovery of solvents • removal of ions from solution, asin demineralization of water • fractionation by selective adsorption of gases,vapours from gases, vapors from vapors and liquids from liquids and many otherapplications.
Extraction- It refers to a separation of the constituents of a liquid solution bycontact with another insoluble liquid. The liquid which is added to the solution tobring about the extraction is known as the solvent. The solution which is to beextracted is called the feed. The solvent-richen product of the operation is calledthe extract and the residual liquid from which the solute is separated is called theraffinate. The separation of aromatics from kerosene based fuel oils, theproduction of fuels in the nuclear industry and the separation of penicillin fromfermentation mixtures are examples of techniques of extraction.
Leaching • Leaching is a process of mass transfer that occurs by extracting a substance from a solid material that has come into contact with a liquid. • In leaching, the liquid is very important as it facilitates the ability to remove (or extract) a given substance from a solid matrix (i.e. material). • Someexamples of leaching operations are oilseed extraction, extraction of sugar beetswith hot water and extraction of medicinal compounds from plant roots, leaves and stems.
Distillation-It is an operation whereby a liquid mixture of miscible and volatilesubstances is separated into individual components or into group of componentsby partial vaporization. The separation of a mixture of methanol and water into itscomponents, and separation of petroleum crude into gasoline, kerosene, fuel oiland lubricating stock are examples of distillation process.
Humidification and dehumidification- In humidification, the enrichment ofvapour content in a gas stream takes place by passing the gas over a liquid. Thetransfer of water from the liquid phase to the gas phase of a mixture of air andwater vapour is a widespread application of humidification. Dehumidificationinvolves the transfer of water vapour from the gas phase to the liquid phase. Removal of water vapour from air by condensation of a cold surface andcondensation of carbon tetrachloride out of a stream of nitrogen are examples of dehumidification.
Drying and Evaporation- Drying refers to removal of relatively small amountof water or other liquid from a solid material whereas evaporation refers to theremoval of relatively large amount of water from solutions. In evaporation the wateris removed as vapour at its boiling point. Drying involves the removal of water attemperature below the boiling point by circulating air or some other carrier gas over the material.
Differential Equations • Conservation of mass in a control volume: • Or, in – out + accumulation – reaction = 0
For in – out, • in x-dir, • in y-dir, • in z-dir, • For accumulation,
For reaction at rate rA, • Summing the terms and divide by DxDyDz, • with control volume approaching 0,
We have the continuity equation for component A, written as general form: • For binary system, • but • and
So by conservation of mass, • Written as substantial derivative, • For species A,
In molar terms, • For the mixture, • And for stoichiometric reaction,
Processes Involving Mass Transfer • Gas-liquid mass transfer: • Several mass-transfer theories have been proposed to explain the mechanism of gas transfer: • The two-film theory (simple and commonly used) • The penetration model (more theoretical (complex)) • The surface-renewal model (more theoretical (complex)) • The Two-Film Theory: • The theory is based on a physical model in which two films exist at the gas-liquid interface. • Two conditions can exist, (a) “absorption”: gas is transferred from the gas phase to the liquid phase and (b) “desorption”: gas is transferred from the liquid phase into the gas phase.
Flux from gas phase into interface: • rg= kg (PG – Pi) • Flux from interface into liquid phase: • rL= kL (Ci– CL) • r = kg (PG – Pi) = kL (Ci– CL) eq(4-122) • Because it is difficult to measure kLand kg at interface, the overall coefficient KG and KL can be used: • r = KL (CS – CL) eq(4-124) • From eq (4-122) and (4-124) • r=KL (CS – CL)=kg (PG – Pi)=kL (Ci– CL) • From Henery’s law at interface: • PG = H CS and Pi = H Ci Where: CS = conc. at interface in equilibrium with partial pressure in the bulk gas phase CL = conc. In the bulk liquid phase
The overall driving force (CS – CL) can be written as: • (CS – CL) = (CS – Ci) + (Ci –CL) • Substituting for PG and Pi in eq (4-122) and combining eq (4-125) and (4-124), the following is obtained where the liquid film controls mass transfer: • If mass transfer is controlled by the gas film: Where: CS = conc. at interface in equilibrium with partial pressure in the bulk gas phase CL = conc. In the bulk liquid phase
To estimate the flux from gas to liquid phase: • Where; r= rate of mass transfer per unit area • Where; = rate of mass transfer per unit volume • = volumetric mass transfer coefficient • = area of mass transfer per unit volume • Using eq(4-130) to describe the mass transfer through the surface of a basin: • Integrating between the limits of C=Co and C=Ct and t=0 and t=t, • (Example 4-10)
Introduction to Process Selection • Process selection involves detailed evaluation of the various factors that must be considered when evaluating unit operations and processes to meet the treatment objectives. • The purpose of the process analysis is to select the most suitable unit operations and processes and the optimal operational criteria. • Important factors in process selection: (Table 4-11) • Process Applicability. The ability of the process to get the job done in terms of eliminating the pollutant. Reflects directly upon the skill and experience of the design engineer. Resources include: data from operating installations; published information in technical journals, manuals of practice (MOPs) from the WPCF; process manuals from EPA and lab and pilot plant studies. • Applicable Flow Range and Variability. For example, stabilization ponds are not suitable for extremely large flowrates, if the flow variation is too great, equalization may be required. • Wastewater Characteristics. Affect the type of processes to be used: physical, chemical, biological. • Many others. Climate, sludge processing, complexity.
MASS TRANSFER THEORIES There are three types of theories in mass transfer coefficients. Film theories Penetration theories Surface –renewal theories
Film Theory • The simplest conceptualization of the gas-liquid transfer process is attributed to Nernst (1904). • Nernst postulated that near the interface there exists a stagnant film . This stagnant film is hypothetical since we really don't know the details of the velocity profile near the interface. • Basic concept – the resistance to diffusion can be considered equivalent to that in stagnant film of a certain thickness • Often used as a basis for complex problems of multicomponent diffusion or diffusion plus chemical reaction.
Mass transfer occurs by molecular diffusion through a fluid layer at phase boundary (solid wall). Beyond this film, concentration is homogeneous and is CAb. • Mass transfer through the film occurs at steady state. • Flux is low and mass transfer occurs at low concentration. Hence,
Integrating Equation (3.55) for the following boundary conditions: CA=CAi when Z=0 CA=CAb when Z=δ We have now:
In this film transport is governed essentially by molecular diffusion. Therefore, Fick's law describes flux through the film.
If the thickness of the stagnant film is given by dn then the gradient can be approximated by: Cb and Ci are concentrations in the bulk and at the interface, respectively.
At steady-state if there are no reactions in the stagnant film there will be no accumulation in the film (Assume that D = constant) -- therefore the gradient must be linear and the approximation is appropriate. And:
Calculation of Ci is done by assuming that equilibrium (Henry's Law) is attained instantly at the interface. (i.e., use Henry's law based on the bulk concentration of the other bulk phase.) Of course this assumes that the other phase doesn't have a "film". This problem will be addressed later. So for the moment: (if the film side is liquid and the opposite side is the gas phase).
A problem with the model is that the effective diffusion coefficient is seldom constant since some turbulence does enter the film area. So the concentration profile in the film looks more like:
Penetration and Surface Renewal Models Most of the industrial processes of mass transfer is unsteady state process. In such cases, the contact time between phases is too short to achieve a stationary state. This non stationary phenomenon is not generally taken into account by the film model. More realistic models of the process have been proposed by Higbie (1935, penetration model) and by Danckwerts ( 1951, surface renewal model). In these models bulk fluid packets (eddies) work their way to the interface from the bulk solution. While at the interface they attempt to equilibrate with the other phase under non-steady state conditions. No film concepts need be invoked. The concentration profile in each eddy ( packet) is determined by the molecular diffusion dominated advective-diffusion equation:
Basic assumptions of the penetration theory are as follows: 1) Unsteady state mass transfer occurs to a liquid element so long it is in contact with the bubbles or other phase 2) Equilibrium exists at gas-liquid interface 3) Each of liquid elements stays in contact with the gas for same period of time Assumption: no advection within the eddy
The solution to this governing equation depends, of course, on boundary conditions. In the Higbie penetration model it is assumed that the eddy does not remain at the surface long enough to affect concentration at the bottom of the eddy ( z = zb). In other words the eddy behaves as a semi-infinite slab. Where C (@ z = zb ) = Cb. Also C (@ z = 0) = Ci .
The boundary conditions are: t = 0, Z > 0 : c = cAb and t > 0, Z = 0 : c = cAi.
Solving the equation with these boundary conditions and then solving for the gradient at z = 0 to get the flux at z = 0 and then finding the average flux over the time the eddy spends on the surface yields the following: q = average time at surface (a constant for a given mixing level). The average mass transfer coefficient during a time interval tcis then obtained by integrating Equation (3.61) as
For the mass transfer in liquid phase, Danckwert (1951) modified the Higbie’s penetration theorywith the surface renewal model. He stated that a portion of the mass transfer surface is replaced with a new surface by the motion of eddies near the surface and proposed the following assumptions: 1) The liquid elements at the interface are being randomly swapped by fresh elements from bulk 2) At any moment, each of the liquid elements at the surface has the same probability of being substituted by fresh element 3) Unsteady state mass transfer takes place to an element during its stay at the interface. s = surface renewal rate (again, a function of mixing level in bulk phase).
Comparison of the models: Higbie and Danckwert's models both predict that J is proportional to D0.5 where the Nernst film model predicts that J is proportional to D. Actual observations show that J is proportional to something in between, D0.5 -1 . There are more complicated models which may fit the experimental data better, but we don't need to invoke them at this time.
Mass transfer coefficients To simplify calculations we usually define a mass transfer coefficient for either the liquid or gas phase as klor kg(dimensions = L/t).