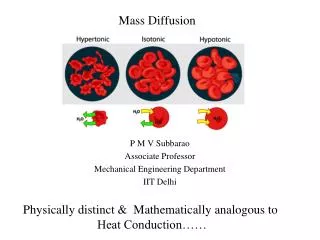
Diffusion Mass Transfer
Diffusion Mass Transfer. Chapter 14 Sections 14.1 through 14.7. Lecture 21. Physical Origins and Rate Equations Mass Transfer in Nonstationary Media Conservation Equation and Diffusion through Stationary Media Diffusion and Concentrations at Interfaces

Diffusion Mass Transfer
E N D
Presentation Transcript
Diffusion Mass Transfer Chapter 14 Sections 14.1 through 14.7 Lecture 21
Physical Origins and Rate Equations • Mass Transfer in Nonstationary Media • Conservation Equation and Diffusion through Stationary Media • Diffusion and Concentrations at Interfaces • Diffusion with Homogenous Reactions • Transient Diffusion
5. Transient Diffusion For 1-D diffusion of species A through a planar medium of A &B without Homogeneous reaction under transient conditions: Two geometries: symmetric planar and semi-infinite media Approach: apply the solutions for transient heat conduction
Transient Diffusion in Planar Medium CA(x,0)=CA,i CA,s CA,s L -L x*=x/L i.c.: CA(x,0) = CA,i b.c.-1: b.c.-2: CA(L,t) = CA,s
CA(x,0)=CA,i CA,s CA,s L -L x*=x/L Transient Diffusion i.c.: b.c.’s:
Transient Diffusion Approximate Solution (Figures 5S.1, 5S.2, 5S.3 in page W12-W15) ζn are positive roots of transcendental equation:
Transient Diffusion Approximate Solution for Fom > 0.2 C1 and ζ1 are listed in Table 5.1, page 301.
Transient Diffusion in Semi-infinite Medium i.c.: CA(x,0) = CA,i b.c.-1: CA(0,t) = CA,s b.c.-2: CA(,t) = CA,i
Transient Diffusion In Semi-infinite Medium
Transient Diffusion in Semi-Infinite Medium x I.C.: t=0, CA(x, 0) = CA0, for all x B.C.-1: x=0, CA(0, t) = CAs, for all t>0 B.C.-1: x→∞, CA(∞, t) = CA0, for all t
Transient Diffusion in Semi-Infinite Medium Let θ = CA-CA0, I.C.: t=0, θ(x, 0) = 0, for all x B.C.-1: x=0, θ(0, t) = CAs-CA0 , for all t>0 B.C.-1: x→∞, θ(∞, t) = 0, for all t
Transient Diffusion in Semi-Infinite Medium θ(x, 0) = 0 θ(0, t) = CAs-CA0 θ(∞, t) = 0 • Methods for solving this 2nd order PDE: • Combination of variables: • Separation of variables: CA(x, t)= f(x) g(t) • Laplace transfer: Crank’s Book • Fourier series analysis: Deen’s Book, Chapter 4.
Transient Diffusion in Semi-Infinite Medium Let Θ =(CA-CAs)/(CA0-CAS) I.C.: t=0, Θ(x, 0) = 0, for all 0≤x≤L B.C.-1: x=0, Θ(0, t) = 1, for all t>0 B.C.-1: x=L, Θ(∞, t) = 0, for all t>0
Transient Diffusion in Semi-Infinite Medium θ(x, 0) = 0 θ(0, t) = CAs-CA0 θ(∞, t) = 0
Example 1 A long nickel bar with a diameter of 5 cm has been stored in a hydrogen-rich environment at 358 K and 300 kPa for a long time, and thus it contains hydrogen gas throughout uniformly. Now the bar is taken into a well-ventilated area so that the hydrogen concentration at the outer surface remains at almost zero at all times. Determine how long it will take for the hydrogen concentration at the center of the bar to drop by half. The diffusion coefficient of hydrogen in the nickel bar at the room temperature of 298 K can be taken to be DAB = 1.2 x 10-12 m2/s, and the solubility of H2 in nickel is 0.00901 kmol/m3bar.
Example 1 Known:Long cylinder with known H2 concentration at given conditions and ambient conditions Find: Time required for H2 concentration in the center of the cylinder to drop by 50% Properties: DAB = 1.2 x 10-12 m2/s, S =0.00901 kmol/m3bar Schematic: Well-ventilated area H2 diffusion H2 gas, 358 K, 300 kPa Nickel bar
Example 1 Assumptions: (1) Infinite long cylinder, (2) Initial H2 concentration in the nickel bar is uniform; (3) H2 at surface remains zero at all time; (4) The Fourier number (mass transfer) is large than 0.2. Analysis: This problem is analogous to the one-dimensional transient heat conduction problem in an infinitely long cylinder with specified surface temperature, and thus can be solved accordingly.
Example 1 Noting that 300 kPa = 3 bar, the molar density of hydrogen in the nickel bar before it is taken out of the storage room is: The molar concentration of hydrogen at the center of the bar can be calculated from:
Example 1 The Biot number in this case can be taken to be infinity since the bar is in a well-ventilated area during the transient case. The constants C1 and 1 for the infinite Bi are determined from Table 5.1 to be 1.6018 and 2.4050, respectively. Noting that the concentration of hydrogen at the outer surface is zero, and the concentration of hydrogen at the center of the bar is one half of the initial concentration, the Fourier number, Fom, can be determined from Using the definition of the Fourier number, the time required to drop the concentration of hydrogen by half is determined to be Therefore, it will take years for this nickel bar to be free of hydrogen.
Example 2 A common procedure for increasing the moisture content is to bubble it through a column of water. Assume the air bubbles to be spheres of radius of r0 =1 mm and to be in thermal equilibrium with the water at 25C. How long should the bubbles remain in the water to achieve concentration at center that is 99% of the maximum possible (saturated) concentration? The air is dry when it enters the water.
Example 2 (4). Bim is very large (>100)
Example 3 A large, deep tank filled with still water, initially has a uniform oxygen concentration of 2 g/m3. The surface concentration of the dissolved oxygen is suddenly raised to 7.5 g/m3. Sketch out the profile of dissolved oxygen concentration versus liquid depth z after 36,000 seconds. At the temperature of the tank, the diffusion coefficient of oxygen in the liquid water is 1.5x10-9 m2/s.
Example 3 Known:Initial dissolved O2 concentration and surface concentration. Find: O2 concentration versus liquid depth after 36,000 seconds. Assumptions: (1) 1-D conditions with constant properties. (2) Semi-infinite geometry. (3) No chemical reaction. (4) Stationary media.
Example 3 Analysis: The conservation of species equation applies to O2 in the liquid water at time t and liquid water depth z:
Example 3 I.C.: t=0, ρO2 (x, 0) = 2 g/m3 B.C.-1: x=0, ρ O2 (0, t) = 7.5 g/m3 B.C.-2: x→∞ρ O2 (∞, t) = 2 g/m3 The solution to the partial differential equation with above IC and BCs are:
Example 3 Lecture 21