CONDUCTION
CONDUCTION. Conduction. Kinetic Energy: Kinetic energy is energy of motion. When an object moves fast, it has lots of kinetic energy . Kinetic Energy. Heat: Heat is a form of Kinetic (motion) energy. For example, molecules of hot air move faster than molecules of cold air. . HEAT.


CONDUCTION
E N D
Presentation Transcript
CONDUCTION Conduction
Kinetic Energy: Kinetic energy is energy of motion. When an object moves fast, it has lots of kinetic energy. Kinetic Energy
Heat: Heat is a form of Kinetic (motion) energy. For example, molecules of hot air move faster than molecules of cold air. HEAT
Fahrenheit Celsius Boiling point of water Freezing point of water Temperature: Temperature is a measure of the average speed of the atoms or molecules. Fahrenheit Scale: On this scale 32 is the freezing temperature of water, and 212 is the boiling point. Celsius Scale: On this scale 0 is the freezing temperature of water, and 100 is the boiling point.
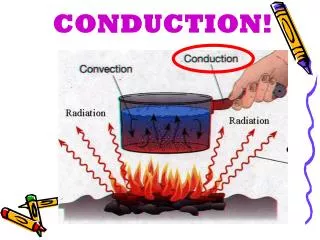
Conduction • Conduction happens mainly in solids. • Heated atoms vibrate and strike neighboring atoms • Energy, or Heat, travels from one atom to the next. Definition: The transfer of heat through molecular activity
Hot (lots of vibration) Cold (not much vibration) Heat travels along the rod. Heat energy always travels in a direction from the hot to the cold.
Back to our Example: ICE COLD LEMONADE Which one is correct? a. The coldness of the ice moves to the lemonade b. The heat from the lemonade moves to the ice c. The coldness and heat move back and forth again and again until the lemonade is cooled d. Other explanation not included here:____________________________