Conduction
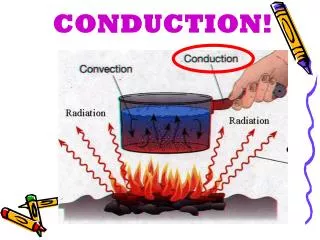
Transfer of heat Heat is a form of energy. It always flows from one place to another, that is from hot to cold places. Heat flows in 3 ways: 1 ) Conduction 2 ) Convection 3 ) Radiation Heat can move through a solid, liquid, gas or vacuum. Conduction.


Conduction
E N D
Presentation Transcript
Transfer of heatHeat is a form of energy. It always flows from one place to another, that is from hot to cold places. Heat flows in 3 ways:1) Conduction2) Convection 3) RadiationHeat can move through a solid, liquid, gas or vacuum.
Conduction • Conductionis the heat transfer through a material medium from places of higher temperature to places of lower temperature. • Conduction is the main method of heat transfer in solids. • Molecules in solids are not free to move from place to place.
What is GOOD CONDUCTOR?The material that conducts heat faster and is a bad insulator.Metals are good conductors of heat.Examples:
What is BAD or POOR CONDUCTOR? • The material that conducts heat slower and is a good insulator. • Non-metals are poor conductors of heat. • Examples:
►The diagram below shows that metal is a good conductor (heat spreads quickly through the base of the pan, warming the food rapidly). The Wooden handle is a bad conductor (heat spreads slowly through the wooden handle so it feels cooler when it is held).
Molecular explanation for conduction Why metals are better conductors than non-metals? In metals Metals are better conductors of heat than non-metals because they contain free electrons. Due to these free electrons, metals are also good conductor of electricity. So, heat is transferred from the hot end to the cold end with the help of these free electrons. When the end P is heated, the free electrons there move faster, gaining more kinetic energy and move farther towards Q carrying the energy with them. As a result, heat energy is transferred from one place to another both by the vibrations of molecules as well as by the movement of free electrons.
Diagram below shows heat is conducted quickly by moving electrons:
In non-metals and solids Non-metals/solids do not have free electrons. This is why they do not conduct heat as well as metals. When the end P of a solid/non-metal is heated, the molecules at P vibrate faster and gain kinetic energy. As a result, the neighbouring molecules are affected to vibrate faster. Finally, the molecules at the end Q also vibrate faster. In this way, heat is transferred from P to Q by conduction.
Diagram below shows heat is conducted slowly by atoms bumping into each other:
Why liquids and gases are poor conductors of heat? The molecules in a liquid are further apart than those in a solid. The molecules of a gas are even further apart than those of a solid and a liquid. Therefore, it is more difficult to transfer the heat energy or movement of the molecules to other molecules for liquids and gases. However, heat can still move through them by convection.
►Diagram below shows molecules of solids, liquids and gases:
Convection What is CONVECTION? Convection is the heat transfer from one place to another by the movement of the liquid or gas itself. Convection is the main method of heat transfer in liquids and gases. The molecules in liquids and gases are free to move from place to place.
Molecular explanation for convection What happen to the molecules of liquids or gases when they are heated? In liquids When a liquid is heated, it expands, becomes less dense and floats upwards. The cold and denser liquid moves down to replace it. This in turn gets heated up. This movement of water is called a convection current.
In gases Convection occurs more readily in gases than in liquids as they expand much more than liquids when their temperature rises.
Radiation What is RADIATION? Radiation is the heat transfer by means of electromagnetic waves or infrared radiation through a vacuum without involving a material medium. The infrared radiation is absorbed by surrounding objects, raising their temperature. Radiation does not depends on the presence of molecules as it can pass through a vacuum. For example, radiation from the sun passes through millions kilometres of space to reach the earth. Heat from the sun is not able to reach the earth by convection or conduction as there is no vacuum in outer space.
Good and Bad Emitters When an object radiates heat, it is said to emit radiation. Different surfaces emit heat differently. Surfaces that are rough and black are good emitters. Surfaces that are shiny and white are bad emitters.
Good and Bad Absorbers When a body's temperature rises, it is said to absorb radiation. Different surfaces absorb heat differently. Surfaces that are rough and black are the good absorbers. Surfaces that are smooth, shiny or white surfaces are the bad absorbers.