Essential Titration Tips for Accurate Acid-Base Concentration Calculations
DESCRIPTION
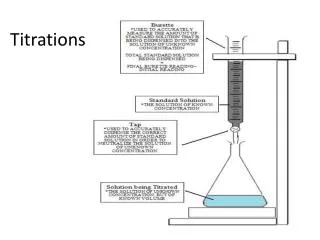
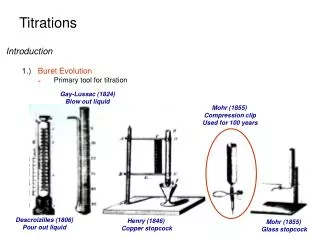
This guide outlines the essential steps for conducting titrations, focusing on the proper techniques for accurately determining the concentration of an acid solution. The procedure begins with rinsing and filling a burette with a basic solution, followed by pipetting the acid into a flask. Careful addition of the base to the acid is crucial, while recording the volume needed for a color change at the endpoint. Finally, calculations for pH and acid concentration are explained, ensuring clarity and precision in results.
1 / 9
Télécharger la présentation 

Essential Titration Tips for Accurate Acid-Base Concentration Calculations
An Image/Link below is provided (as is) to download presentation
Download Policy: Content on the Website is provided to you AS IS for your information and personal use and may not be sold / licensed / shared on other websites without getting consent from its author.
Content is provided to you AS IS for your information and personal use only.
Download presentation by click this link.
While downloading, if for some reason you are not able to download a presentation, the publisher may have deleted the file from their server.
During download, if you can't get a presentation, the file might be deleted by the publisher.
E N D
Presentation Transcript
Step 6: Record volume of base required to change color of acid
Step 8: Calculate the concentration of the acid M = moles/litre Moles base = Moles acid (end point)
More Related