Titrations
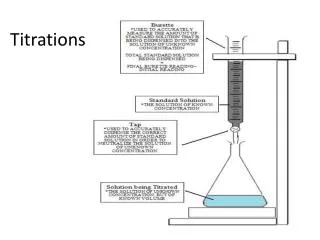
Titrations. Titration is a volumetric procedure for determining the concentration of an unknown species by adding a carefully measured volume of a known species that reacts with the unknown .

Titrations
E N D
Presentation Transcript
Titration is a volumetric procedure for determining the concentration of an unknown species by adding a carefully measured volume of a known species that reacts with the unknown. Theequivalence point in a titrationoccurs when the requirements of the balanced equation for reaction are exactly satisfied. An indicator is used to find the end-point pf the titration. The end-point is the point where the indicator changes colour. This may not be at the equivalence point
For an acid base titration the equivalence point is the point where neutralisation occurs. Indicators must have the following properties : a) the color change must be sharp. i.e. the indicator must change colour with the addition of one drop of solution from the burette. b) change colour as close to the equivalence point as possible c) Give a distinct colour change
Titration Curves Strong Acid-Strong Base Titrations The equivalence point of the titration is the point at which exactly enough titrant has been added to react with all of the substance being titrated with no titrant left over. In other words, at the equivalence point, the number of moles of titrant added so far corresponds exactly to the number of moles of substance being titrated according to the reaction stoichiometry.
Titrations Involving a Weak Acid or Weak Base • There are three major differences between this curve (in blue) and the one we saw before (in black): • 1. The weak-acid solution has a higher initial pH. • 2. The pH rises more rapidly at the start, but less rapidly near the equivalence point. • 3. The pH at the equivalence point does not equal 7.00. • POINT OF EMPHASIS : The equivalence point for a weak acid-strong base titration has a pH > 7.00.
Weak Base / Strong Acid 1. The weak-base solution has a lower initial pH. 2. The pH drops more rapidly at the start, but less rapidly near the equivalence point. 3. The pH at the equivalence point does not equal 7.00. POINT OF EMPHASIS : The equivalence point for a weak base-strong acid titration has a pH < 7.00.
Choosing an indicator In the strong acid titration, both indicators begin to change colour at the equivalence point (50 mL of base) so both work equally well. In the weak acid titration, thymol blue changes colour at the equivalence point, but methyl red begins to change colour after only 15mL of base are added, which is far from the equivalence point, illustrating the importance of choosing an appropriate indicator.
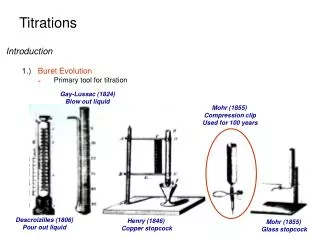
Primary Standards A primary standard is a reagent that is extremely pure, stable, has no waters of hydration, and has a high molecular weight. Some primary standards for titration of acids: sodium carbonate: mol wt. = 105.99 g/mol tris-(hydroxymethyl)aminomethane (TRIS or THAM): mol wt. = 121.14 g/mol Some primary standards for titration of bases: potassium hydrogen phthalate (KHP): mol wt. = 204.23 g/mol potassium hydrogen iodate: mol wt. = 389.92 g/mol Some primary standards for redox titrations: potassium dichromate mol wt. = 294.19 g/mol
Secondary Standards A secondary standard is a standard that is prepared in the laboratory for a specific analysis. It is usually standardized against a primary standard. Some secondary standards for titration of acids Sodium hydroxide Potassium hydroxide
Standardisation Calculations Sodium carbonate (anhydrous) is used as a primary standard. A solution of sodium carbonate of concentration 0.100 mol/L is used to standardise a solution of hydrochloric acid. 25.0 ml of the standard solution requires 35 ml of the acid for neutralisation. Find the concentration of the acid. Na2CO3 + 2HCl 2 NaCl + CO2 + H2O • 1 mole of Na2CO3 neutralises 2 moles of HCl • No of moles of Na2CO3 in 25 ml =0.1 x 25 = 2.5 x 10 -3 mol • 1000 • No of moles HCl in 35 ml = 2 x 2.5 x 10 -3 = 5.0 x 10 -3 mol • Conc of the acid = 5.0 x 10 -3 x 1000 = 0.143 mol/L 35