Titrations
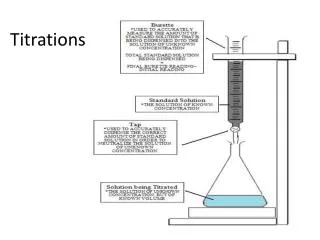
Titrations. Volumetric analysis Procedures in which we measure the volume of reagent needed to react with an analyte Titration Increments of reagent solution ( titrant ) are added to analyte until reaction is complete. - Usually using a buret

Titrations
E N D
Presentation Transcript
Titrations • Volumetric analysis • Procedures in which we measure the volume of reagent needed to react with an analyte Titration • Increments of reagent solution (titrant) are added to analyte until reaction is complete. - Usually using a buret • Calculate quantity of analyte from the amount of titrant added. • Requires large equilibrium constant(Thermodynamic) • Requires rapid reaction • (kinetic) • aA + tT → products • A: analyte • T: titrant http:\\asadipour.kmu.ac.ir 41 slides
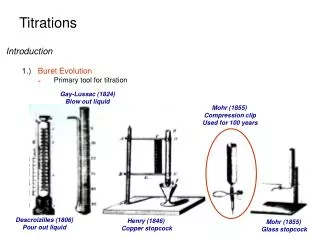
Titrations • BuretEvolution Gay-Lussac (1824) Blow out liquid Mohr (1855) Compression clip Used for 100 years Descroizilles (1806) Pour out liquid Henry (1846) Copper stopcock Mohr (1855) Glass stopcock http:\\asadipour.kmu.ac.ir 41 slides
Type of Titrations based on Measuring Techniques • Volumetric titrimetry: Measuring the volume of a solution of a known concentration (e.g., mol/L) that is needed to react completely with the analyte. • Gravimetric (weight) titrimetry: Measuring the mass of a solution of a known concentration (e.g., mol/kg)that is needed to react completely with the analyte. • Coulometrictitrimetry: Measuring total charge (current x time) to complete the redox reaction, then estimating analyte concentration by the moles of electron transferred. http:\\asadipour.kmu.ac.ir 41 slides
Type of Titrations based on Chemical Reactions • Acid-Base Titrations, example: • H+ + OH–→ H2O • Precipitation Titrations, example: • Ag+(aq) + Cl–(aq)→AgCl(s) • Redox Titrations: • 5 H2O2 + 2 MnO4–+ H+→ 5 O2 + 2 Mn2+ + 8H2O • Complexometric Titrations, example: • EDTA + Ca2+→ (Ca–EDTA)2+ http:\\asadipour.kmu.ac.ir 41 slides
Type of Titration Curves V. = volume http:\\asadipour.kmu.ac.ir 41 slides
Formality • Molarity (V & W) Molality Normality %W/W %W/V %V/V part per thousand (ppt) - X's 1000 parts per million (ppm) - X's 106 parts per billion (ppb) - X's 109 Expressing concentration http:\\asadipour.kmu.ac.ir 41 slides
# Eqgtitrant = # Eqganalyte (V*N)titrant=(V*N)analyte # Eqgtitrant = (V*N)titrant #molestitrant=(V*M)titrant #molesanalyte=(V*M)analyte relationship between titrant and analyte http:\\asadipour.kmu.ac.ir 41 slides
Standardization: The process by which the concentration of a reagent is determined by reaction with a known quantity of a second reagent • Primary standard: The reagent which is ready to be weighted and used prepare a solution with known concentration (standard). Requirements of primary reagent are: - Known stoichiometric composition and reaction - High purity - Nonhygroscopic - Chemically stable both in solid and solution - High MW or FW • Secondary standard: A standard solution which is standardized against a primary standard. http:\\asadipour.kmu.ac.ir 41 slides
Standardization • Required when a non-primary titrant is used • - Prepare titrant with approximately the desired concentration • - Use it to titrate a primary standard • - Determine the concentration of the titrant • - Titration Standardization titrantknown concentration titrantunknown concentration analyteunknown concentration analyteknown concentration http:\\asadipour.kmu.ac.ir 41 slides
Standardization of 0.1 M NaOH N1V1=N2V2 1-selection the PS (e.g. KHP) 2-wheing the PS 10*0.1=mg/204.1 213.8 3-making solution 4-addind suitable indicator 5-titration 9.1ml 6-calculation 9.1*n=213.8/204.1 n=0.115 http:\\asadipour.kmu.ac.ir 41 slides
Blank Titration: Titration procedure is carried out without analyte (e.g., a distilled water sample). It is used to correct titration error. • Back titration: A titration in which a (known) excess reagent is added to a solution to react with the analyte. The excess reagent remaining after its reaction with the analyte, is determined by a titration. http:\\asadipour.kmu.ac.ir 41 slides
Standardization Example: To standardizing a KMnO4 stock solution, the primary standard of 9.1129 g Na2C2O4 is dissolved in 250.0 mL volumetric flask. 10.00 mL of the Na2C2O4 solution require 48.36 mL of KMnO4 to reach the titration end point. What is the molarity (M) of MnO4– stock solution? (FW Na2C2O4 134.0) Solution: 5C2O42–(aq) + 2MnO4–(aq) + 16H+(aq) → 10CO2(g) + Mn2+(aq) + 8H2O(l) Ans http:\\asadipour.kmu.ac.ir 41 slides
Unknown Analysis with a Blank Correction Example: A 0.2865 g sample of an iron ore is dissolved in acid, and the iron is converted entirely to Fe2+. To titrate the resulting solution, 0.02653 L of 0.02250 M KMnO4 is required. Also a blank titration require 0.00008 L of KMnO4 solution. What is the % Fe (w/w) in the ore? (AW Fe 55.847) MnO4–(aq) + 5Fe2+ + 8H+(aq) → Mn2+(aq) + 5Fe3+ + 4H2O(l) http:\\asadipour.kmu.ac.ir 41 slides
Back Titration • 1)Add excess of one standard reagent (known concentration) • 2)Titrate excess standard reagent to determine how much is left - Add Fe2+ to determine the amount of MnO4- that did not react with oxalic acid - Differences is related to amount of analyte - Useful if better/easier to detect endpoint MnO4–(aq)+ 5Fe2+ + 8H+(aq) → Mn2+(aq) + 5Fe3+ + 4H2O(l) http:\\asadipour.kmu.ac.ir 41 slides
Example: The arsenic in 1.010 g sample was pretreated to H3AsO4(aq) by suitable treatment. The 40.00 mL of 0.06222 M AgNO3 was added to the sample solution forming Ag3AsO4(s): The excess Ag+ was titrated with 10.76 mL of 0.1000 M KSCN. The reaction was: Calculate the percent (w/w) As2O3(s) (fw 197.84 g/mol) in the sample. Back Titration http:\\asadipour.kmu.ac.ir 41 slides
Back Titration In a back titration analysis of HCO3-, 25 mL of a bicarbonate solution is reacted with 25.00 mL of 0.100 M NaOH. The excess NaOH was titrated with 0.100 M HCl. This required 14.82 mL. What is the concentration of bicarbonate in solution? NaOH + HCO3-→ Na+ + CO3- + H2O NaOH + HCl→NaCl + H2O http:\\asadipour.kmu.ac.ir 41 slides
Analyte Oxalic acid (colorless) Titrant (purple) (colorless) (colorless) Equivalence point • Quantity of added titrant is the exact amount necessary for stoichiometric reaction with the analyte - Ideal theoretical result Equivalence point occurs when 2 moles of MnO4-is added to 5 moles of Oxalic acid http:\\asadipour.kmu.ac.ir 41 slides
Analyte Oxalic acid (colorless) Titrant (purple) (colorless) (colorless) End point • Occurs from the addition of a slight excess of titrant • Marked by a sudden change in the physical property of the solution • -Change in color, pH, voltage, current, absorbance of light. -End point ≈equivalence point After equivalence point occurs, excess MnO4- turns solution purple Endpoint http:\\asadipour.kmu.ac.ir 41 slides
Titration Error • Differencebetween endpoint and equivalence point • Corrected by a blank titration • 1) repeat procedure without analyte • 2) Determine amount of titrant needed to observe change • 3) subtract blank volume from titration http:\\asadipour.kmu.ac.ir 41 slides
Calculation of ascorbic acid in Vitamin C tablet: Standardization: Suppose 29.41 mL of I3- solution is required to react with 0.1970 g of pure ascorbic acid, what is the molarity of the I3- solution? Analysis of Unknown: A vitamin C tablet containing ascorbic acid plus an inert binder was ground to a powder, and 0.4242g was titrated by 31.63 mL of I3-. Find the weight percent of ascorbic acid in the tablet. • Starch is used as an indicator: • starch + I3- starch-I3- complex ascorbic acid was oxidized with I3-: 1 mole ascorbic acid 1 mole I3- http:\\asadipour.kmu.ac.ir 41 slides
Example: A solid mixture weighing 1.372 g containing only sodium carbonate (Na2CO3, FW 105.99) and sodium bicarbonate (NaHCO3, FW 84.01) require 29.11 mL of 0.7344 M HCl for complete titration: Find the mass of each component of the mixture. Titration of a Mixture http:\\asadipour.kmu.ac.ir 41 slides
Ans Ans http:\\asadipour.kmu.ac.ir 41 slides
Direct and back (indirect) titration of Aspirin http:\\asadipour.kmu.ac.ir 41 slides
Precipitation Titrations A titration in which the reaction between the analyteand titrant involves a precipitation. Ag+(aq)+ Cl–(aq)AgCl(s) AgCl(s) Ag+(aq) + Cl–(aq)Ksp = 1.8×10–10 s = [Ag+]=[Cl–] [Ag+]=[Cl–]=1.35x10–5 pAg = 4.89 pCl = 4.89 http:\\asadipour.kmu.ac.ir 41 slides
pCl pAg Titration curve of 50.0 mL of 0.0500 M Cl– with 0.100 M Ag+ http:\\asadipour.kmu.ac.ir 41 slides
Example: For the titration of 50.0 mL of 0.0500 M Cl– with 0.100 M Ag+. The reaction is: Ag+(aq) + Cl–(aq)AgCl(s) K = 1/Ksp = 1/(1.8×10–10) = 5.6 x 109 Find pAg and pCl of Ag+ solution added 0 mL (b) 10.0 mL (c) 25.0 mL (d) 35.0 mL Solution: (a) 0 mL Ag+ added (At beginning) [Ag+] = 0, pAg can not be calculated. [Cl–] = 0.0500, pCl = 1.30 (b) 10 mL Ag+ added (Before Ve) N1V1=N2V2 50*0.05=0.1*V2 In eq point, 25 ml √Ksp=[Ag+]=[Cl-]=1.34*10-5 (d) 35 mL Ag+ added (After Ve) http:\\asadipour.kmu.ac.ir 41 slides
Diluting effect of the titration curves 25.00 mL 0.1000 M I– titrated with 0.05000 M Ag+ 25.00 mL 0.01000 M I– titrated with 0.005000 M Ag+ 25.00 mL 0.001000 M I– titrated with 0.0005000 M Ag+ http:\\asadipour.kmu.ac.ir 41 slides
Ksp effect of the titration curves 25.00 mL 0.1000 M halide (X–) titrated with 0.05000 M Ag+ http:\\asadipour.kmu.ac.ir 41 slides
Titration of a mixture (uncertainty concerned) • 40.00 mL of 0.0502 M KI + 0.0500 M KCl, titrated with 0.0845 M Ag+ • 20.00 mL of 0.1004 M KI titrated with 0.0845 M Ag+ http:\\asadipour.kmu.ac.ir 41 slides
Example: A 25.00 mL solution containing Br– and Cl– was titrated with 0.03333 M AgNO3. Ksp(AgBr)=5x10–13, Ksp(AgCl)=1.8x10–10. • Which analyte is precipitated first? • The first end point was observed at 15.55 mL. Find the concentration of the first that precipitated (Br– or Cl–?). • The second end point was observed at 42.23 mL. Find the concentration of the second that precipitated (Br– or Cl–?). Solution: (a) Ag+(aq) + Br–(aq) AgBr(s) K = 1/Ksp(AgBr) = 2x1012 Ag+(aq) + Cl–(aq) AgCl(s) K = 1/Ksp(AgCl) = 5.6x109 Ans: AgBr precipitated first http:\\asadipour.kmu.ac.ir 41 slides
Argentometric Titration • Define Argentometric Titration: • A precipitation titration in which Ag+ is the titrant. • Argentometric Titration classified by types of • End-point detection: – Volhard method: A colored complex (back titration) – Fajans method: An adsorbed/colored indicator – Mohr method: A colored precipitate http:\\asadipour.kmu.ac.ir 41 slides
Mohr method The Mohr method was first published in 1855 as a method for chloride analysis. ·In the precipitation of chloride by silver ion, chromate ion (CrO42) is used as an indicator in the formation of Ag2CrO4, a reddish-brown precipitate formed when excess Ag+ is present. Ag+ + ClAgCl(s) white precipitate 2Ag+ + CrO42Ag2CrO4(s) red precipitate CrO42 Ksp= 1.8 x 10-10 (S = 1.34 x 10-5 M) Ksp= 1.2 x 10-12 (s = 6.7 x 10-5M) http:\\asadipour.kmu.ac.ir 41 slides
The titrations are performed only in neutral or slightly basic medium to prevent silver hydroxide formation (at pH > 10). 2Ag+ + 2OH2AgOH(s) Ag2O(s) + H2O precipitate to prevent chromic acid formation (at pH < 7). CrO42 + H3O+ HCrO4 + H2O 2 CrO42 + 2 H3O+ H2CrO4+ H2O http:\\asadipour.kmu.ac.ir 41 slides
Volhard METHOD Back titration for determination of Cl-..First published in 1874. Reactions: Ag+ + ClAgCl(s) Ksp = 1.82 x 10-10 (excess)white precipitate SCN +Ag+AgSCN(s) Ksp= 1.1 x 10-12 titrant white precipitate SCN + Fe3+FeSCN2+ Kf = 1.4 x 10+2 Indicator red complex H+, Fe3+ http:\\asadipour.kmu.ac.ir 41 slides
The titration is usually done in acidic pH medium to prevent precipitation of iron hydroxides, Fe(OH)3. Fe3+ +3(OH)- ⇄ Fe(OH)3Ksp=1*10-39 If [Fe3+]=0.001 M pH=????? http:\\asadipour.kmu.ac.ir 41 slides
Since SAgSCN <SAgCl, equilibrium will shift to the right causing a negative error for the chloride analysis. Ag+ + ClAgCl(s) Ksp = 1.82 x 10-10 SCN +Ag+AgSCN(s) Ksp = 1.1 x 10-12 SCN- + AgClAgSCN + Cl- To eliminate this error, AgCl must be filtered or add nitrobenzene before titrating with thiocyanate; nitrobenzene will form an oily layer on the surface of the AgCl precipitate, thus preventing its reaction with thiocyanate. http:\\asadipour.kmu.ac.ir 41 slides
Fajans Method: An adsorbed/colored indicator. Titrating Cl– and adding dichlorofluorosceinas indicator: Before Ve (Cl– excess) Greenish yellow solution After Ve (Ag+ excess) http:\\asadipour.kmu.ac.ir 41 slides
5) Applications of argentometric titrations: http:\\asadipour.kmu.ac.ir 41 slides