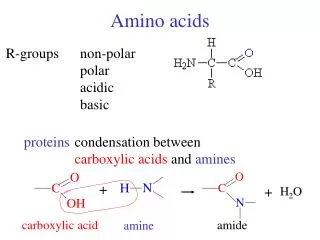
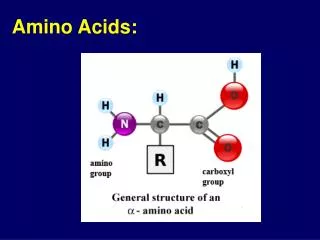
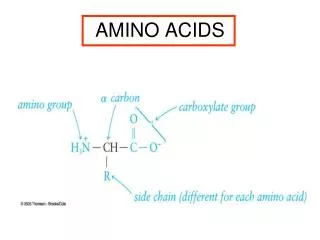
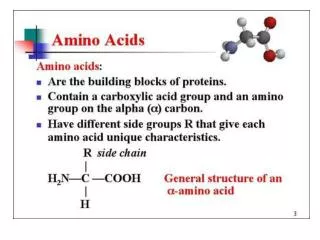
Amino acids
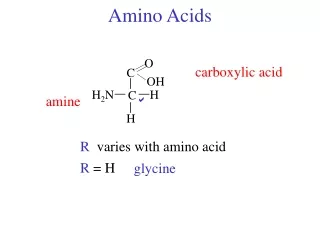
Amino acids. Met dank aan Dr. Detke. Most amino acids are chiral. All naturally occuring amino acids except Gly are the L isomer L & D amino acids are stereoisomers. Most amino acids are zwitterions. amino acids are zwitterions at neutral pH (contains negative and positive charged groups).


Amino acids
E N D
Presentation Transcript
Amino acids • Met dank aan Dr. Detke
Most amino acids are chiral • All naturally occuring amino acids except Gly are the L isomer • L & D amino acids are stereoisomers
Most amino acids are zwitterions • amino acids are zwitterions at neutral pH (contains negative and positive charged groups)
Glycine non-chiral smallest most flexible in polypeptides less hydrophobic Alanine chiral next smallest less hydrophobic Small aliphatic amino acids
Val, Leu, Ile Highly hydrophobic branched side-chains Other aliphatic amino acids
Proline imino acid rigid ring structure puts kinks in polypeptides Yet another aliphatic amino acid
Aromatic amino acids • Phenylalanine • Absorbs UV • Very hydrophobic • Tyrosine • Absorbs UV • less hydrophobic • H-bonds • OH can ionize • OH can be modified • Tryptophan • Really absorbs UV! • less hydrophobic • H-bonds
Methionine highly hydrophobic initiator amino acid Cysteine less hydrophobic forms disulfide bonds SH group can ionize Sulfur-containing amino acids
Disulfide bond formation is reversible Disulfide bonds form in proteins and help stabilize their structures Oxidation of cysteine to cystine
Ser, Thr less hydrophobic H-bonding potential can be modified Thr is an isostere of Val Alcoholic amino acids
Asp, Glu highly hydrophilic negatively charged at neutral pH Acidic amino acids
Highly hydrophilic Lys and Arg are positively charged at neutral pH His is partially charged at neutral pH Basic amino acids
Asn, Gln highly hydrophilic H-bonding potential isosteres of Asp and Glu Amide-containing amino acids
The peptide bond is planar Fig. 4.6
Aspartame – a dipeptide 200X Sweeter than Sucrose !!! aspartylphenylalanine methyl ester