Carbon Compounds and Chemical Bonds
920 likes | 1.13k Vues
Carbon Compounds and Chemical Bonds. Chapter 1. ORGANIC CHEMISTRY. STUDY . OF. CARBON. COMPOUNDS. CONTAINING. Compounds from Nature Synthetic compounds: invented by organic chemists and prepared in their laboratories. Friedrich Woehler’s urea synthesis. 1828.

Carbon Compounds and Chemical Bonds
E N D
Presentation Transcript
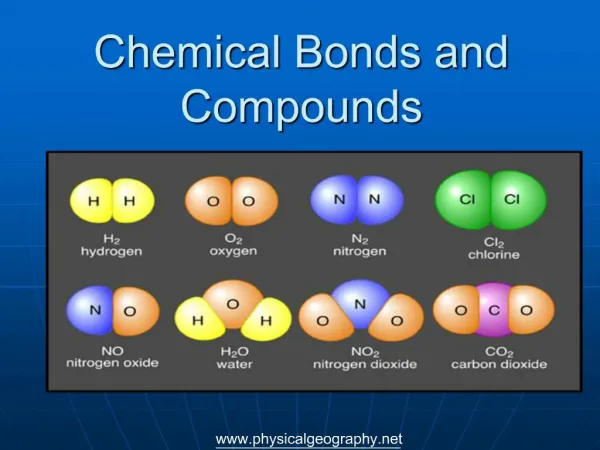
Carbon Compounds and Chemical Bonds Chapter 1
ORGANIC CHEMISTRY STUDY OF CARBON COMPOUNDS CONTAINING Compounds from Nature Synthetic compounds: invented by organic chemists and prepared in their laboratories Friedrich Woehler’s urea synthesis 1828 Ammonium isocyanate + heat ------> urea NH4CNO NH2CONH2 1828 “I have been able to make urea without aid of kidney of man or dog”.
Some organic chemicals • Medicines • Active Pharmaceutical Ingredients • Excipients DNA Fuels Materials Essential oils Pigments
AND WITH ITSELF No limit
Electronic Structure of Atoms • Structure of atoms • a small dense nucleus, diameter 10-14 - 10-15 m, which contains positively charged protons, neutrons and most of the mass of the atom • extranuclear space, diameter 10-10 m, which contains negatively charged electrons
Notice: ones orbital in each principal shell threep orbitals in the second shell (and in higher ones) fived orbitals in the third shell (and in higher ones)
Rules for Electron Configurations Capacities of shells (n) and subshells (l)
Electronic Structure of Atoms • Electrons are confined to regions of space called principle energy levels (shells) • each shell can hold 2n2 electrons (n = 1, 2, 3, 4......)
Electronic Structure of Atoms • Shells are divided into subshells called orbitals, which are designated by the letters s, p, d,........ • s (one per shell) • p (set of three per shell 2 and higher) • d (set of five per shell 3 and higher) .....
Electronic Structure of Atoms • Rule 1:orbitals fill from lowest energy to highest energy • Rule 2: only two electrons per orbital, spins must be paired • Rule 3: for a set of orbitals with the same energy, add one electron in each before a second is added in any one
Li Na K Ca Sr Ba “Periodic” Behavior of Elements Flame tests: elements with low first ionization energies are excited in a flame, and often emit in the visible region of the spectrum Atoms emit energy when electrons fall from higher to lower energy states
Electronic Structure of Atoms • The pairing of electron spins
Lewis Structures • For Nitrogen atom: • Valence shell of Nitrogen= 3 • Number of valence electrons of Nitrogen = 5 • Gilbert N. Lewis • Valence shell: the outermost electron shell of an atom • Valence electrons: electrons in the valence shell of an atom; these electrons are used in forming chemical bonds • Lewis structure • the symbol of the atom represents the nucleus and all inner shell electrons • dots represent valence electrons
Lewis Structures • Lewis structures for elements 1-18 of the Periodic Table • For Nitrogen atom: • Valence shell of Nitrogen= 3 • Number of valence electrons= 5
Lewis Model of Bonding • Atoms bond together so that each atom in the bond acquires the electron configuration of the noble gas nearest it in atomic number • an atom that gains electrons becomes an anion • an atom that loses electrons becomes a cation • Ionic bond: a chemical bond resulting from the electrostatic attraction of an anion and a cation • Covalent bond: a chemical bond resulting from two atoms sharing one or more pairs of electrons • We classify chemical bonds as ionic, polar covalent, and nonpolar covalent based on the difference in electronegativity between the atoms
Electronegativity • Electronegativity: a measure of the force of an atom’s attraction for the electrons it shares in a chemical bond with another atom • Pauling scale • increases from left to right within a period • increases from bottom to top in a group
Electronegativity • Electronegativity of atoms (Pauling scale)
Electronegativity and chemical bonding Electronegativity • Example: NaCl • Na = 0.8, Cl = 3.0 • Difference is 2.2, so • this is an ionic bond!
Coulomb’s Law “The energy of interaction between a pair of ions is proportional to the product of their charges, divided by the distance between their centers”
Covalent Bonding Forces • Electron – electron • repulsive forces • Proton – proton • repulsive forces • Electron – proton • attractive forces
Bond Length Diagram Scientists can determine the internuclear distances that correspond to the lowest energy states of molecules Net repulsion Net attraction http://ch301.cm.utexas.edu/simulations/bond-strength/BondStrength.swf
Bond Length and Energy Bonds between elements become shorter and stronger as multiplicity increases
Covalent Bonds • A covalent bond forms when electron pairs are shared between two atoms whose difference in electronegativity is 1.9 or less • an example is the formation of a covalent bond between two hydrogen atoms • the shared pair of electrons completes the valence shell of each hydrogen.
Polar Covalent Bonds • In a polar covalent bond • the more electronegative atom has a partial negative charge, indicated by the symbol d- • the less electronegative atom has a partial positive charge, indicated by the symbol d+ • in an electron density model • red indicates a region of high electron density • blue indicates a region of low electron density
ammonia and formaldehyde are polar molecules • acetylene is a nonpolar molecule Polar and Nonpolar Molecules
Carbon– Intro and Review • Atomic Structure • Atoms – made up of protons, neutrons, electrons • Isotopes – same # protons; different # neutrons • Electronic Structure • Electrons • determine structure • give rise to bonding • behave like waves • orbitals (s, p)
Electron Probabilitiesand the 1s Orbital The 1s orbital looks very much like a fuzzy ball, that is, the orbital has spherical symmetry The electrons are more concentrated near the center Spherical symmetry; probability of finding the electron is the same in each direction. The electron cloud doesn’t “end” here … … the electron just spends very little time farther out.
The region near the nucleus is separated from the outer region by a spherical node - a spherical shell in which the electron probability is zero Electron Probabilitiesand the 2s Orbital The 2s orbital has two regions of high electron probability, both being spherical
Subshell filling order ... Rules for Electron Configurations Each subshell must be filled before moving to the next level 1s22s22p63s23p6 ... 1s < 2s < 2p < 3s < 3p < 4s < 3d < 4p < 5s < 4d < 5p < 6s
The most stable arrangement of electrons in subshells is the one with the greatest number of parallel spins (Hund’s rule). Ne 1s22s22p6 F 1s22s22p5 O 1s22s22p4 N 1s22s22p3 C 1s22s22p2
Periodic Relationships • The valence shellis the outermost occupied shell • The period number = principal quantum number, n, of the electrons in the valence shell
Atomic Orbitals • 1s – 1st orbital • s type (spherical) • 1s, 2s, 3s
2s orbital (spherical) Atomic Orbitals
2p orbital Atomic Orbitals • p (2p, 3p…) • 3 orbitals oriented perpendicular to each other • have node (region of 0 e- density) • nodal plane
Atomic Orbitals • p (2p, 3p…) • 3 orbitals oriented perpendicular to each other • have node (region of 0 e- density) • nodal plane • shape • dumbbell
Electronic Configuration of Atoms • Aufbau • Fill lowest energy orbital 1st • Hund’s Rule • 1 e- into each orbital of = energy • Pauli Exclusion Principle • Electrons in the same orbital are spin paired Chapter 1
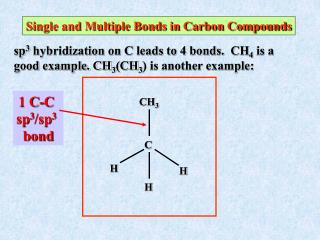
WHY DO HYBRIDS ?? • 1. Electron pair repulsions are minimized (= lower energy) • 2. Stronger bonds (= lower energy) are formed • 3. Hybrids have better directionality for forming bonds
Shapes of Atomic Orbitals • All sorbitals have the shape of a sphere, with its center at the nucleus • of the sorbitals, a 1s orbital is the smallest, a 2s orbital is larger, and a 3s orbital is larger still
Shapes of Atomic Orbitals • A p orbital consists of two lobes arranged in a straight line with the center at the nucleus
Orbital Overlap Model • A covalent bond forms when a portion of an atomic orbital of one atom overlaps a portion of an atomic orbital of another atom • in forming the covalent bond in H-H, for example, there is overlap of the 1s orbitals of each hydrogen