Understanding the Classification and Properties of Matter
150 likes | 325 Vues
Learn about different phases of matter, physical and chemical changes, mixtures, pure substances, and laws governing matter composition. Discover methods to separate mixtures and identify chemical changes.

Understanding the Classification and Properties of Matter
E N D
Presentation Transcript
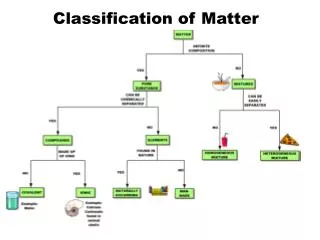
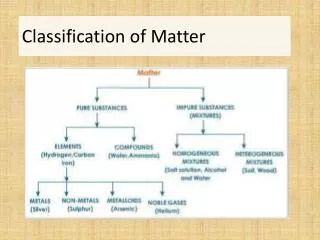
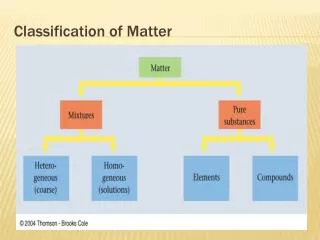
Classification of Matter • Matter – anything with mass and volume
Phase Differences Solid – definite volume and shape; particles packed in fixed positions. Liquid – definite volume but indefinite shape; particles close together but not in fixed positions Gas – neither definite volume nor definite shape; particles are at great distances from one another Plasma – high temperature, ionized phase of matter as found on the sun.
Condense Freeze Evaporate Melt Gas Liquid Solid
Chemical vs. Physical Change Physical change – changes form, not composition - all changes in phase are physical Chemical change – change that produces a new substance(s) with different properties
Mixtures • Made up of two substances together but not bonded • Heterogeneous- mixture is not the same from place to place. • Homogeneous- same composition throughout (also called solutions)
Mixtures Can Be Separated by Physical changes • Distillation • Filtration • Chromotography
Pure Substances Compounds – substance with a constant composition - can be broken down into smaller elements be chemical processes Elements – substance that cannot be decomposed into simpler substances by chemical or physical means - composed of smallest component of matter (Atoms)
Chemical Changes can be Identified by Observation Change in color Change in temperature Formation of a gas Release of light Formation of a precipitate
Law of Conservation of Mass In any chemical or physical change – matter (and its mass) is neither created nor destroyed
Law of Definite Composition A given compound always contains the same proportion of elements by mass Example: Water has a mass ratio of hydrogen to oxygen of 1:8. What will the mass of oxygen be in a sample of water that contains 3g of hydrogen?
Law of Multiple Proportions When two elements form a series of compounds, the ratios of the masses of the second element that combine with 1g of the first element can always be reduced to small whole numbers