Classification of Matter in Chemistry
160 likes | 207 Vues
Explore the classification of matter into mixtures and pure substances, including elements and compounds. Learn about extensive and intensive properties, and differentiate between homogeneous and heterogeneous mixtures.

Classification of Matter in Chemistry
E N D
Presentation Transcript
What is classification? • Classify the following things into two classes: • Dog • Cat • Dolphin • Whale • Tuna • Shark • Tiger • Human
Classification in Chemistry • Chemistry focuses on matter and its interactions • We rely on knowing how matter is classified to tell us about its properties
Types of Properties • Extensive Properties: a property that changes when the size of the sample changes • Ex: mass volume
Types of Properties • Intensive Properties: Properties that remain constant independent of the amount of sample present • Ex: melting point hardness density
Self Check • Determine if each of the following is an extensive or intensive property • Smell • Density • Length • Luster • Malleability
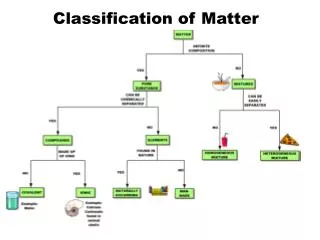
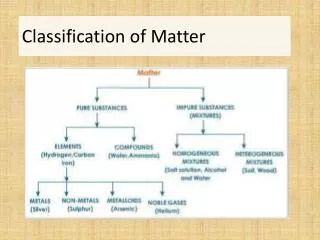
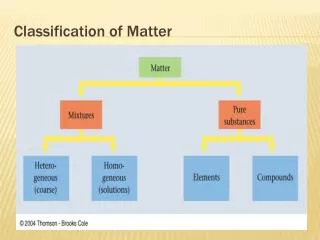
Matter • Anything that has mass, and takes up space! • Everything is matter! • Matter can be broken down into two classes: • Mixtures • Pure substances
Pure Substance • Form of matter that cannot be separated by physical means • Can be broken down into: • Elements • Compounds
Elements • A pure substances that cannot be further decomposed (broken down) by ordinary chemical methods • Elements are made of all of the same type of atoms • These are what make up the periodic table • Examples • Oxygen (O) • Gold (Au) • Iron (Fe)
Compounds • Compound • can be decomposed (broken down) into two or more simpler substances (elements) by ordinary chemical means • Compounds are made of multiple elements • Properties of a compound are usually different from the properties of the elements that make it up • Examples (usually given formula) • Water (H2O) • Sodium Chloride (NaCl)
Classes of Matter cont. • Mixture • Two or more substances physically mixed together • A mixture generally has the same properties as the substances that make it up • Can be broken down into: • Homogenous Mixture • Heterogeneous Mixture
Mixtures Ctd • Homogeneous Mixture • Homo = same • Also called a solution • Homogeneous mixture has the same properties throughout • Parts are not visible • Examples? • Salt water • Kool Aid
Mixtures Ctd • Heterogeneous Mixture • Hetero = different • Heterogeneous mixture has different properties throughout • Parts are visible • Examples? • Fruit salad • Vegetable Soup
Matter Flow Chart • Make a flow chart, starting with matter, that helps to explain how matter is classified • Terms that must be on map (include an example of each as well): • Matter, mixture, pure substance, element, compound, homogenous mixture, heterogeneous mixture, solution
Classify the Following using Flow Chart • Lead (Pb) • Iced Tea • Milk • Dirt • Silver (Ag) • Rocky Road Ice Cream • Snickers Bar • Gatorade • Hydrogen Peroxide (H202) • Nachos • Water Vapor (H20) • Gravel • Wood • Sugar (C6H12O6)
Classify the Following using Flow Chart • Nickel (Ni) • Chocolate Milk • Pizza • Beach • Salt (NaCl) • Trail Mix • Glue • Salsa • Coffee • Spaghetti • Water • Vegetable Oil • Gas (CH4) • Sugar (C6H12O6)