Sedimentation and Sedimentary Rocks
740 likes | 960 Vues
Sedimentation and Sedimentary Rocks. GLY 2010 - Summer 2014 - Lecture 10. Sediment. Unconsolidated material that accumulates at the earth’s surface Minerals and organic remains of plants and animals are the major components of soil. Sedimentary Rock.

Sedimentation and Sedimentary Rocks
E N D
Presentation Transcript
Sedimentation and Sedimentary Rocks GLY 2010 - Summer 2014 - Lecture 10
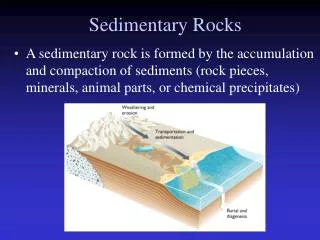
Sediment • Unconsolidated material that accumulates at the earth’s surface • Minerals and organic remains of plants and animals are the major components of soil
Sedimentary Rock • As sediment accumulates, pressure, and often temperature, increases • Changes occur which convert the sediment from unconsolidated form to a consolidated form, sedimentary rock • Sedimentary rock makes up 5% of the crust of the earth, but accounts for 75% of the rock exposed at the surface
Importance of Sedimentary Rock • Provide clues to the earth’s past – examples: • Including erosion of mountain ranges • Transgressions of the sea over the land
Fossils • Often contain fossils, which provide clues to: • Types of life living in the past • The environment they lived in
Types of Sediment • Detrital • Chemical • Biogenic
Sediment Transport • Water, glaciers, or wind moves the sediment from high elevations to lower elevations, where it may accumulate • Annual transport of detrital sediments to the oceans is about 10 billion tons • During transportation, sediment size is often reduced
Transport of Sediment By rivers By glaciers
Formation of Detrital Rock • Deposition • Sorting • Shape
Lithification • Literally means creation of stone • Involves three possible processes • Compaction • Cementation • Recrystallization
Compaction • Fragments will be compacted by the weight of accumulating sediment • Air and water are expelled from spaces between grains
Cementation • Dissolved substances in water may precipitate solids which act as cements
Recrystallization • Unstable minerals may reorganize due to heat, pressure, and fluid interaction into more stable minerals • Process must occur at low temperatures • Ex: Aragonite Calcite
Sedimentary Structures • Bedding - sediments are ordinarily deposited in horizontal units called beds • Graded bedding • Cross-bedding
Cross-bedding, Zion National Park Photos: Duncan Heron
Principle of Original Horizontality • Most sediments settle through bodies of water • They will be deposited in horizontal, or very nearly horizontal, layers • Beds which are not horizontal have often had their position changed by post-depositional processes
Right Side Up • Beds may occasionally be completely overturned, so we need ways to tell if beds are right side up • Selected indicators: • Ripple Marks • Mudcracks • Raindrop impressions • Salt crystals
Aeolian Ripple Marks • Aeolian (wind) ripples at White Sands, New Mexico (Photo Yamato Sato)
Fossilized Ripple Marks • Parallel ripple marks preserved on a slab of sandstone • The rocks contain the fossilized remains of marine animals - these ripples were formed in shallow sea water by gentle currents • Fossilized ripple marks. Capitol Reef National Park, Utah
Mudcracks Mudcracks form by desiccation of mud or clay
Salt Crystals • If the deposition occurs in the ocean, and the water is quite saline, salt crystals may precipitate and settle on the sediment
Size Range of Detrital Particles • Clay < 0.004 millimeters • Silt 0.004 to 0.063 millimeters • Sand 0.063 to 2 millimeters • Granule 2 to 4 millimeters • Pebble 4 to 64 millimeters • Cobble 64 to 256 millimeters • Boulder >256 millimeters
Sediment Types • Mud is composed of clay or silt • Sand is composed exclusively of sand sized particles • Gravel includes granules, pebbles, cobbles, and boulders
Formation of Chemical Sedimentary Rocks • Precipitation is the process of converting materials dissolved in a fluid (water or air) to another form • Water dissolved in the atmosphere may precipitate as rain, or as some solid form such as snow, sleet, hail, etc • Solids dissolved in water precipitate as solids
Saturated Solution • In order for precipitation to occur, the fluid must be saturated • A saturated solution holds as much as it can of a particular substance • A fluid may be saturated with respect to one substance (e.g. lime) and undersaturated with respect to another substance (e.g. halite)
Chemical Precipitation • Solutions which are saturated, or slightly supersaturated, may spontaneously form crystals, which settle in the solution
Biochemical Precipitation • Some organisms have the ability to concentrate an unsaturated solution internally to the point where precipitation occurs This fossiliferous limestone contains carbonate shells, produced by biochemical precipitation
Evaporites • When saline solutions (sea-water) evaporate, a series of substances precipitate in a definite sequence, from the least to the most soluble • Sequence is: • Lime – a carbonate • Gypsum, a sulfate, precipitates second • Halite, common table salt, is next • Potassium and magnesium salts are last
Chemical Sedimentary Rocks • Limestone is composed of calcium carbonate, CaCO3 • Dolostone is composed of dolomite, CaMg(CO3)2
Oolitic Limestone • In warm, saturated, and highly energetic environments, lime may precipitate around tiny bits of suspended matter • They stick together to form an oolitic limestone, such as the Miami oolite formation
Biochemical Sedimentary Rock • Coral reefs • Coquina
Ancient Marine Reef • El Capitan reef, Guadalupe Mountains National Park, formed as part of a very large reef complex during the Permian period