Chapter 5 Cell disintegration and extraction techniques
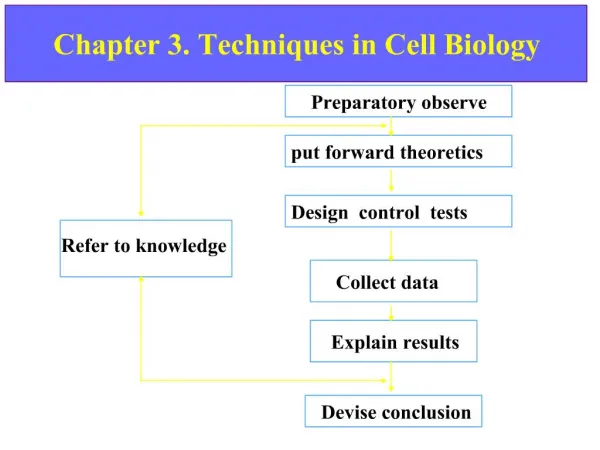
Chapter 5 Cell disintegration and extraction techniques. Overview. To get the intracellular product the disintegration of cells is need

Chapter 5 Cell disintegration and extraction techniques
E N D
Presentation Transcript
Chapter 5 Cell disintegration and extraction techniques
Overview • To get the intracellular product • the disintegration of cells is need The methods used to break cells depend largely on the fragility of the cells ex) animal cells: burst by osmotic shock, freeze/thaw, enzyme digestion(lipase, protease), toluen plant tissue: pectinase and cellulase treatment microbial cells: lysozyme treatment To achieve a good yield,1.minimize the number of steps2. choose appropriate disruption methods (a) can a given disruptor be used for a particular cell type? (b) which is the best method of extracting a product?
Key questions • Stability of the released protein • Location of target protein within the cell • The yield and kinetics of the process • Continuous or batch disruption • The need to consider subsequent steps • Assesing the extent of disruption • Marker substance for cell disruption • Containment of the process-avoid release of harzardous intracellular products into envronment • Scale-up-cost, volume, sample viscosity
1.Stability of the released protein • The disruption methods can impose great physical and chemical stress ex) heat generated by mechnical disruption may result in Nz denaturation activation of proteolytic enzyme can degrade target Nz • Consideration: minimize the stress condition 3.The yield and kinetics of the process • Yield-the quantity of enzyme released/unit starting materials • Specific activity-the amount of enzyme(unit)/ released protein(g) • Factors affecting the yield of enzyme (a) location of product with in the cell (b) degree of disintegration (c) extent of denaturation of the product during disruption • Disruption rate can be modelled by “First-order process” R=RM(1-e-kt) : time-dependentconsider the “optimun disruption time”
In(RS/RL) *18μω2(ρP-ρL)dP2 t= 5. The need to consider subsequent steps • After disruption, clarification step is need for subsequent purification step • usually, through centrifugation in the laboratory, high g force on larger scale, clarifying cell lysate is difficult whether a laboratory centrifuge is capable of sedimenting cell debris t: the centifugal time RS:radii from centrifuge tube head to liquid surface RL:radii from centrifuge tube head to bottom μ:liquid velocity ρP:particle density ρL:liquid dencity ω:angular velocity =2π(rpm)/60 dP:mean diameter of cell debris • Mechanical disruption methods are not needed chemicals that intetfere with subsequent purification step
6. Assesing the extent of disruption • Use a marker substance (estimate total intracellular proteins) • specific activity could decrease during disruption because of releasing of non-product proteins and denaturation of enzyme during disruption 7. Marker substance for cell disruptionto determine the degree of cell disruption, marker techniques are used 1. Biological: visible cell counting 2. Physical: measuring the Vol. of intact cells, O.D., viscosity of the sample 3. Chemical: measuring the protein concentration
Methods of disruption • Slow the rate of enzyme damage caused by proteas(add PMSF in eukariotic cell) • Prevent thermal denaturation 4℃ • Pre-treatment of material • General procedure notes • Mixers and blenders • Coarse grinding: pestle and mortar • Fine grinding:the bead mill 10. Osmotic shock • Homogenization 11. Lytic enzymes • Ultrasonication 12. Chemical treatment • Heat shock 13. Detergents • Freezing and thawing 14. Slovents
1. Pre-treatment of material To increase the degree of cellular disruption Combine two(or more) methods for example, freeze-thaw step or pre-treatment of solvents or detergents before mechnical disruption
2. General procedure notes Table 1.The main cell disruption methods Disadvantage:Cells are not extensively distintegrated • Methods needing specialist equipment (a) mixer and blender (b) coarse grinding with pestle and mortar (c) fine grinding in a bead mill (d) homogenization (e) ultrasonication • Methods using non-specialist equipment (a) freezing and thawing (b) osmotic shock (c) chaotrophic agents (d) detergents (e) solvents (f) enzyme lysis incubation timeoperational parameters(conc.of lysing chemicals, bead size, conc., agitator speed for bead mill, pressure for homogenization, Vol. Of sample for ultrasonication) Disadvantage: addition of extra chemicals to the system may interfere with downstream processing
3. Mixers and blenders • Grind cells coarsely • Use the buffer containing inhibitors, reducing agents(see protocol 2) 4. Coarse grinding: pestle and mortar • Useful for disruption of tissue samples • Samples were grinded to fine powder under liquid nitrogen • Nacessary to maintain the frozen state 5. Fine grinding: the bead mill • Useful for disruption of micro-orgarnisms • Bead mills should have cooling jackets because of heat generation during disruption • Optional parameters: bead size, bead volume, agitator speed, milling time
6. Homogenization • Hand-held piston/plunger deviceanimal cells are easily disrupted, but very inefficient • High-pressure homogenizersuitable for large scale operationprinciples; • sample narrow orfice cell crusing cell breakingusually, increasing the pressure increase the amount and rate of protein release according to k=k’Pn(first-order reaction, p98) High pressure pumping High-pressure homogenizer Mechanical homogenizer
7. Ultrasonication • Very vigorous process, results in complete solubilization • Principles; the vibrating titanium probe creat cavitiescollaps of the cavitiespressure changes and shear foreces which cause cell disruption • Problem: heat-generation(cause thermal denaturation, alteration of Nz activity impossible to adjust the power input • The release curve for protein is usually first-order(Fig. 4), but could be affected by power input, sample volume • Very useful for fragmentaion of cellular DNA related with increaseing of viscosity
Non-mechanical disruption methods Heat shock • Consider the thermal denaturation. Leading to loss of Nz activity • Useful for purification of heat-stable proteins(e.g.ubiquitin) Freezing and thawing • Very simple, but suitable for cells without a cell wall • Repeating of freezing and thawing may cause denaturation of proteins Osmotic shock Lytic enzymes – bacterial disruption with lysozme useful for Gram positive cells Chemical treatment ·dvantage: the cell will be left substantially intact· chemicals must bo compatible with further downstrea process · chelating agents(sequest divalent cation)and chaotropic agents(weaken hydrophobin interaction) Detergents-increase protein solubility Slovents – dosen’t inactivate the enzyme products toluene, ether, isoamylalcohol, chloroform
Colclusions: choice of methods • The mechanical methods of cell disruption have the widest application for laboratory and pilot scale disruption • Homogenization has proved an effective large scale process • Chemical methods are, generally, cell/product specific and thus, not applicable to all system