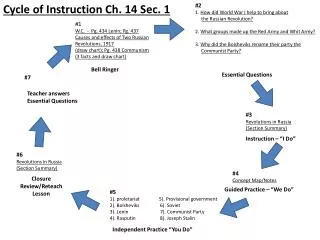
BELL RINGER
BELL RINGER. Explain, in terms of particles and energy, the different states of matter. Matter & Energy. MATTER & ENERGY. anything that has mass and volume. MATTER -. ENERGY -. the ability to do work. NO. Mixture. Pure Substance. Can it be physically separated?.

BELL RINGER
E N D
Presentation Transcript
BELL RINGER Explain, in terms of particles and energy, the different states of matter.
Matter & Energy
MATTER & ENERGY anything that has mass and volume MATTER - ENERGY - the ability to do work
NO Mixture Pure Substance Can it be physically separated? Can it be chemically decomposed? Is the composition uniform? YES Yes Yes No No Matter Homogeneous Mixture Heterogeneous Mixture Compound Element
Examples • Graphite - • Pepper - • Sugar(sucrose) - • Paint - • Milk - element heterogeneous compound mixture Homogeneous mixture
BELL RINGER YES, you have to write down the chart • Classify each type of mixture. • Determine the volume of Fe fillings used to produce mixture 2.
½ day Notes
Ne N2 SO3 Homogeneous Mixture
Physical vs. Chemical • Physical Change - • changes the form of a substance without changing its identity • properties remain the same • Examples • dissolving, changes of phase
Temperature (ºC) Time (sec) BELL RINGER 1.What does C6H12O6 (aq) mean? 2. Looking at the graph on the right, characterize each variable as independent or dependent.
Physical vs. Chemical • Chemical Change - • changes the identity of a substance • products have different properties • ID by color change, bubbles, heat • Examples • rust, burning a log, ice pack
Physical vs. Chemical • Chemical Changes - Mythbusters Mentos Rocket
NO Mixture Pure Substance Can it be physically separated? Can it be chemically decomposed? Is the composition uniform? YES Yes Yes No No Matter Compound Element Homogeneous Mixture Heterogeneous Mixture
1. Suspension Mixtures Made of two or more separate compounds In chemistry, this is usually a solid and a liquid A. Types of mixtures Heterogeneous Largest particles Over 1 µm If left alone, particles will settle Can also filter the mixture to remove particles 2. Colloid Heterogeneous Smaller particles, may be invisible Between 0.001 - 1 µm Mixture is not transparent Particles do not settle. Cannot be filtered
3. Solution Homogeneous Smallest particles Under 0.001 µm Invisible - solution appears transparent Particles cannot be filtered Must distill the solution to remove solids B. Solutions 1. Parts Solute - Smaller of the two materials – usually solid Larger quantity of the two – usually water or liquid Solvent - (aq) Solutions are labeled CaCl2(aq) Calcium chloride dissolved in water 2. Solubility How much solute can go into a solvent
Tyndall Effect • Used to identify a solution, light is reflected by the dissolved particles
NO Mixture Pure Substance Can it be physically separated? Can it be chemically decomposed? Is the composition uniform? YES Yes Yes No No Matter Compound Element Homogeneous Mixture Heterogeneous Mixture suspensions solutions colloids
BELL RINGER Which of the following phase changes are exothermic? Just right down the correct choice! • CO2(s) + heat CO2(g) • NH3(g) NH3(l) + heat • Cu(s)+ heat Cu(l) • Hg(l)+ heat Hg(g)
STATES OF MATTER • Solids • Liquids • Gases • Plasma
Atom Movement What happens to an atom when the kinetic energy changes? Click Here Vibrational Rotational Translational - particles are constantly vibrating - about an axis, they flip over end to end - particles move from place to place
STATES OF MATTER SOLIDS - • very low KE - particles can vibrate but not move • fixed shape & volume • Do Not conform to the container shape • Vibrate only
STATES OF MATTER • Crystalline solid – arranged in a specific pattern such as diamonds, salt, or ice • Amorphous solid – no molecular order as found in charcoal, plastics and glass
STATES OF MATTER LIQUIDS - • low KE - particles can move around but are still close together • variable shape but packed closely together • fixed volume • (incompressible) • Vibrate & Rotate
STATES OF MATTER GASES - • high KE - particles can separate and move throughout the container • Variable shape & volume • Fluid and Compressible • Vibrate, Rotate, Translate
STATES OF MATTER PLASMA - • Very high KE - particles collide with enough energy to break into charged particles (+/-) • variable shape & volume • Stars, fluorescent light bulbs
STATES OF MATTER Animation Very good display of the differences in states of matter
HEAT vs. Temperature Temperature - the measure of an object’s average kinetic energy - the more an object moves, the higher the temperature Heat - flow of energy from a higher temperature object to a lower temperature object
Think Using the following graph, draw a line, representing the temperature of a substance as it is heated constantly starting as a solid all the way through to a gas. Temperature (KE) Time
Heating Curve Temperature (KE) KE PHASE KE PHASE KE Time
BELL RINGER The temperature of a piece of copper with a mass of 95.4 g increases from 25.0°C to 48.0°C when the metal absorbs 849 J of heat. What is the specific heat of copper? q = mcΔT 849J = (23.0 K) (c) (95.4 g) c = 0.388 J/g•K
Heating Curve Temperature (KE) q=mc T q=mHv q=mc T q=mHf m = mass c = specific heat [4.18] q = heat loss/gain q=mc T Time
Heat Energy Problems How much heat energy, in joules, is absorbed by 100. grams of water when it is heated from 20.0ºC to 30.0 ºC? Get the formula from Reference Tables q = mcΔT q = 100.g (4.18J/g•ºC) 10.0ºC q = 4180 J
Heat Energy Problems How much heat energy is absorbed when 35.0g of ice melts to form liquid water at the same temperature? Get the formula from Reference Tables q = mHf q = (35.0 g) (334J/g) q = 11690 J q = 11700 J
Heat Energy Problems How much heat is absorbed when 70.00 g of water is completely vaporized at its boiling point? Get the formula from Reference Tables q = mHv q = (70.0 g) (2260J/g) q = 158200 J
Heat Energy Problems If it takes 273.3 J of energy to condense 19.2 g of a substance, what is the heat of vaporization of a substance Get the formula from Reference Tables q = mHv 273.3J = (19.2 g) (x) Hv = 14.2 J/g
BELL RINGER If it takes 22.0 kj of energy to change 43.2 g of a substance to a liquid, what is its heat of fusion? 509 Joules/gram
Heat of Fusion Lab Remember: The energy to melt the ice only comes from the warm water!!!!!!!!!
BELL RINGER A What Just sketch the graph! B C Lauric Acid is starting as a liquid D Which line segment represents a phase change only? What is the melting point of lauric acid? At which point do the particles of lauric acid have the highest kinetic energy? What phase change occurs during this 10-minute graph?
Separation Techniques Since the components of a mixture are different substances, with at least some physical properties that are unique to each substance, mixtures can be separated by physical means into their components by techniques such as … Filtration Distillation Decanting Chromatography And many others
Mixture of solid and liquid Stirring rod Funnel Filter paper traps solid Filtrate (liquid component of the mixture) Filtration Is the process of removing ‘straining’ a solid, precipitate, from a liquid using a porous paper
Filtration • For separation of substances in different phases • Ex – Air filters, coffee filters, fuel filters • Decanting • Immiscible liquids – separation by differing densities use a separatory funnel
decanting • Pouring a liquid off of a solid
decanting • carefully pouring a solution from a container, leaving the precipitate in the bottom of the container. • Precipitate– solid formed in a solution during a chemical reaction
chromatography • Separation of a mixture based upon bonding preferences or size of molecules • Tie-dye • Pen ink • Chlorophyll • Paternity testing