Understanding the Naming of Acids: A Comprehensive Guide
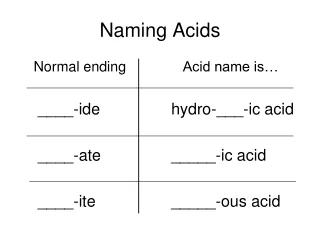
This guide provides an overview of acid naming conventions based on their composition. Acids are homogeneous mixtures producing hydronium ions (H3O+) and are typically expressed in aqueous form. Naming depends on whether the acid contains oxygen. For non-oxy acids, use "hydro" plus the modified element name with “ic acid.” For oxyacid compounds, alter the suffix of the polyatomic anion: “ate” to “ic acid” and “ite” to “ous acid.” Practice exercises help reinforce the naming and formula writing of various acids.

Understanding the Naming of Acids: A Comprehensive Guide
E N D
Presentation Transcript
Acids • Homogeneous mixture that produce hydronium ion (H3O+) • The formulas begin with H and end with (aq) *(aq) = aqueous, meaning “dissolved in water” (Ex) HCl(aq), H2SO4(aq) • Naming depends if the acid has oxygen or not (Ex) HCl(aq), H2SO4(aq)
Naming Acids that Do Not ContainOxygen 1. Read the hydrogen as “hydro” 2. Modify the second’s element name with “ic acid” (Ex) HCl(aq) : hydro HCl(aq) : hydrochloric acid (Ex) H2S(aq): hydrosulfuric acid ** fluoric; iodic, bromic, selenic, cyanic,
Naming Acids that Contain Oxygen • Don’t read the hydrogen • If the polyatomic anion’s name ends with “ate”, change it to “ic acid” (Ex) H2SO4(aq): sulfuric acid 3. If the polyatomic anion’s name ends with “ite”, change it to “ous acid” (Ex) H2SO3(aq): sulfurous acid
Practices • Name the acids. 1. HNO2(aq) 2. H2 CO3(aq) 3. HF(aq)
Practices • Write the formulas 1. nitric acid 2. hypochlorous acid 3. acetic acid 4. hydroselenic acid