Naming Acids
Naming Acids. Haley Dana Chase Mykoal Nikki Maddie Brinda. Oxyacids. Any acid containing hydrogen and an oxyanion oxyanion- a polyatomic ion containing oxygen. Naming Oxyacids. Identify the oxyanion Find the root of the oxyanion Change the suffix Add “acid” to the end.

Naming Acids
E N D
Presentation Transcript
Naming Acids Haley Dana Chase Mykoal Nikki Maddie Brinda
Oxyacids • Any acid containing hydrogen and an oxyanion • oxyanion- a polyatomic ion containing oxygen
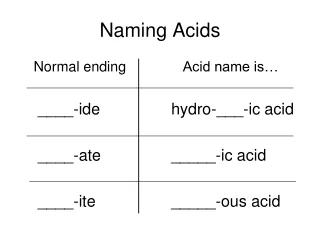
Naming Oxyacids • Identify the oxyanion • Find the root of the oxyanion • Change the suffix • Add “acid” to the end
Example: HNO3 • Identify HNO3 as an Oxyacid • Identify the polyatomic • Nitrate(NO3) • Change the suffix • Nitrate→ Nitric • Add the word “acid” All together… Nitric Acid
Example 2: Carbonic Acid • Identify Carbonic Acid as an oxyacid • Take off the word “acid” • Carbonic • Change the suffix using the chart to identify the polyatomic acid • Carbonic → Carbonate • Use the polyatomic formula • CO3-2 • Add Hydrogen to beginning • H2CO3 • Dont forget to swap and drop!!!
Binary Acids • Contains hydrogen and one other element
Naming Binary Acids • Add prefix- Hydro • Find the root of the second element plus suffix -ic • Add “acid” at the end
Example: HBr • HBr • Add “Hydro” • Root of bromine: Brom- • add suffix: -ic • Add acid at the end All together… Hydrobromic Acid
Example 2 Hydrosulfuric Acid
Strong Acid • An acid that ionizes completely • ionizing: break apart into positive and negative ions • Examples: • Hydrochloric- HCl • Hydrobromic- HBr • Hydroiodic- HI • Perchloric- HClO4 • Nitric- HNO3 • Sulfuric- H2SO4