Molecules
Molecules. Objectives. Write the electron dot structure for an atom. Explain how covalent bonds form molecules. Electron Dot Structures. Dots can be used to represent v/e. Write electron dot structures for K, P, S, and Br. Covalent Bonds. How do non-metal atoms bond together?.

Molecules
E N D
Presentation Transcript
Objectives • Write the electron dot structure for an atom. • Explain how covalent bonds form molecules.
Electron Dot Structures Dots can be used to represent v/e. Write electron dot structures for K, P, S, and Br.
Covalent Bonds How do non-metal atoms bond together? covalent bond: an attraction between non-metal atoms sharing v/e in a molecular orbital Both nuclei are attracted to the molecular orbital. 2e- Example: chlorine gas, Cl2 + + Both nuclei repel each other. This equilibrium establishes the bond length.
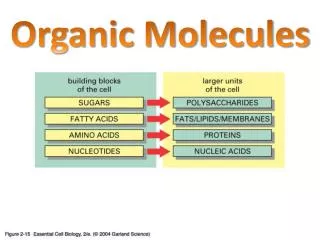
Molecules molecule: group of neutral atoms held together with covalent bonds molecular formula: indicates number and kinds of atoms in a molecule H2O2 O H H O hydrogen peroxide
Objective • Be able to draw a Lewis diagram when given a molecular formula.
Lewis Structures • Draw Lewis structuresfor molecules containing the • following elements: • nitrogen and fluorine • sulfur and chlorine • hydrogen and oxygen (water)
Lewis Structures • Double and triple covalent bonds can also form: • H2CO (double) • ClCN (triple)
Objective • Understand the concept of VSEPR theory. • Use VSEPR theory to determine the shape(s) of a molecule. • Be able to identify common molecular shapes.
VSEPR Theory Valence Shell Electron Pair Repulsion Theory: each pair of electrons (bonding pair or unshared pair) will repel; molecule will adjust shape to maximize the angles between each pair
Molecular Shapes CH4 methane tetrahedral (4 single) NH3 ammonia pyramidal (3 single) bent (2 single) H2S dihydrogen sulfide
Molecular Shapes trignonal planar (2 single, 1 double) H2CO formaldehdye CO2 carbon dioxide linear (2 double) linear (1 single, 1 triple) HCN hydrogen cyanide
Objectives • Be able to determine the polarity of a covalent bond using a table of electronegativities. • Be able to determine the polarity of a molecule based on the shape(s) present and the polarity of the covalent bonds within the molecule. • Understand and apply the concept of molecular symmetry.
Bond Polarity Electrons are not always shared evenly between atoms. 2.2 4.0 Atom with higher electronegativity attracts the e- pair more! H F d+ d- polar bond: unevenly shared covalent bond 3.0 3.0 Br Br non-polar bond: evenly shared covalent bond
Bond Polarity Electronegativity difference determines bond polarity: 0.0 – 0.4 = non-polar covalent (even sharing) 0.5 – 1.9 = polar covalent (uneven sharing) 2.0 – above = ionic bond (e- transfer) What is the polarity of a H – C bond? A bond between H – O? A bond between K – Cl?
Molecular Polarity Bond polarity and molecular shape must be considered when determining the polarity of a molecule. dipole: a polar molecule with polar bonds in which partial charges can be separated d- NH3 d+ H2O d+ d-
Molecular Polarity A molecule with all non-polar bonds is called a non-polar molecule. Hydrocarbons (with just H and C) are always non-polar molecules. Due to symmetry, a molecule with polar bonds can be a non-polar molecule. d- d+ d-
Objectives • Be able to determine the type of intermolecular bonding present in a molecular compound. • Be able to predict the state of a molecular compound based on the type of intermolecular bonding present and the mass of the molecules.
Intermolecular Bonds Covalent bonds form within molecules (polar or non-polar). intermolecular bond: a bond between molecules State of substance determined by strength Non-polar molecules tend to be gaseous because they don’t attract each other. Examples: O2, N2, CO2
London Dispersion Forces London dispersion force: an intermolecular force caused by random distributions of electrons Brief partial charges cause attraction—more electrons causes stronger bonding! Generally, in a non-polar molecule… gas: < 70 e- liquid: 70 e- to 100 e- solid: > 100 e- d-d- d- d+d+ d+ d-d- d-
Dipole Interactions • dipole interaction: an attraction between dipoles that always results in a liquid or solid • London force strength will determine state: • liquid: < 100 e- • solid: > 100 e- d- d+ d+ d- d- d+ d- d+
Hydrogen Bonds • hydrogen bond: a particularly strong dipole interaction • that occurs between molecules containing –OH or –NH • always liquid or solid (depends on strength of London • forces)
Hydrogen Bonds H-bonds occur in both H2O and DNA.
Objectives • Be able to draw a Lewis diagram for a polyatomic ion. • Be able to draw a Lewis diagram for a molecule or ion containing coordinate covalent bonds.
Molecular Names Covalent Compound or Molecular Compound Nomenclature Prefixes 1 mono- (mon-) 2 di- 3 tri- 4 tetra- (tetr-) 5 penta- (pent-) 6 hexa- (hex-) 7 hepta- (hept-) 8 octa- (oct-) 9 nona- (non-) 10 deca- (dec-) • use prefixes that match the number of atoms in the formula • end the second name with “-ide” • never start the first name with “mono-“ • The o or a at the end of a prefix is often dropped when the word following the prefix begins with another vowel; however, it is okay to have an a before the i in iodide
Diatomic Elements • Many elements exist a paired atom molecules • H2, N2, O2, and halogens
Macromolecules macromolecule: a huge molecule C and Si produce macromolecules because each element can form four bonds per atom quartz (SiO2) diamond (C)
Properties of Covalent Compounds • Composed on non-metals only • Covalent bonding—exist as molecules. • Solid, liquid, or gas • Usually non-conductors of heat and electricity
Polyatomic Ions and Coordinate Covalent Bonds (extra-credit) Polyatomic ions are essentially charged molecules (usually with additional electrons). Draw Lewis structures for NO2– and OH– coordinate covalent bond: a bond that forms when one atom donates both electrons to a bond Examples: CO, SO3, CO32–