Testicular Seminoma
540 likes | 886 Vues
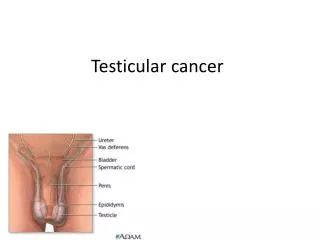
Testicular Seminoma. The Nuts and Bolts of Therapy Matthew Harkenrider, MD Radiation Oncology PGY4. Epidemiology. Accounts for only 1-2% of all cancers in men, but… The most common solid tumor in males between 15 and 35 years of age Estimated 8450 new cases, 350 deaths in US in 2010

Testicular Seminoma
E N D
Presentation Transcript
Testicular Seminoma The Nuts and Bolts of Therapy Matthew Harkenrider, MD Radiation Oncology PGY4
Epidemiology • Accounts for only 1-2% of all cancers in men, but… • The most common solid tumor in males between 15 and 35 years of age • Estimated 8450 new cases, 350 deaths in US in 2010 • Cumulative lifetime risk for a white American male is 0.2% • Seminoma seen more frequently in 25-45 yrs, NSGCT seen more in 15-30 year age group
Etiology • Environmental factors? • Mumps orchitis, immunosuppression following organ transplant, HIV • History of trauma likely leads to testis examination leading to finding unrelated testicular cancers • Prenatal factors including threated miscarriage, excess maternal nausea, C-section • The only definitely associated factors are…
Etiology • Cryptorchidism (undescended testicle) • 5x increased risk (1 in 100) • Orchipexy before puberty lowers risk some • 5-20% occur in contralateral testicle • Mechanism unknown – temperature? trauma? • Previous testicular cancer • Seminoma – 3.6% @ 25 years • Non-seminoma – 8.4% @ 25
Prevention and Early Detection • Screening? • No formal screening recommended • Most found on self exam or by chance • Pt’s with previous h/o Testicular CA should have routine examination
Clinical Evaluation • Most common presentation is a painless testicular mass. About 45% will have some pain • After a mass is detected… • Testicular U/S – shows solid mass • Tumor Markers • AFP is elevated in NSGCT (or mixed with NSGC component), but not in seminoma • β-hCG found in NSGCT and 5-10% of seminoma • LDH 30-80% of seminoma • Radical Inguinal Orchiectomy – tumor staging • CT Abd/Pelvis – nodal staging • CXR – metastatic staging
Pathology • The vast majority (>95%) of testicular cancers are GCT’s • Classified into seminomatous and nonseminomatous for clinical purposes • 60% pure seminoma, 30% NSGCT, 10% mixed • A characteristic chromosome abnormality in GCTs of all histologic types is the 12p isochromosome
Pathology – Seminoma • Well demarcated from normal tissue • Virtually all express placental leukocyte alkaline phospatase (PLAP) • Anaplastic seminoma when >3 mitosis/hpf which is a more aggressive variant
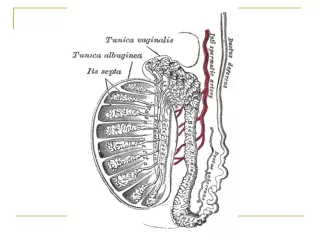
Pathways of Spread • Local extension occurs • into epididymis (T1) • Through tunica vaginalis (T2) • Into spermatic cord (T3) • Into scrotum (T4)
Pathways of Spread • Lymphatic spread • Right testis interaortocaval precaval paraaortic • Left testis paraaortic preaortic interaortocaval • Contralateral involvement found in approx 15% • Pelvic and inguinal involvement occurs in <3% • Supradiaphragmatic involvement can occur via the thoracic duct • Supraclavicular involvement can occur in the setting of relapse
Nodal Staging • N0 – negative • N1 – ≤ 2 cm • N2 – 2.1-5.0 cm • N3 – >5 cm
Staging • Stage I – any T, N0, M0 • Stage II – any T, N1-3, M0 • Stage III – M1
Primary Therapy – Surgery • Inguinal orchiectomy to allow high division of the spermatic cord • Scrotal approach not recommended – can alter lymphatic drainage • May not be essential for patients with bilateral tumors • Orchiectomy alone offers a cure in ~85% of patients with stage I disease
Seminoma • 70-80% of patients will present with clinical stage I disease • 15-20% have infradiaphragmatic node involvement radiologically (Stage II) • Less than 5% have distant disease
Historically… • Stage I Seminoma was treated with inguinal orchiectomy with adjuvant radiation to the paraaortic nodes • Relapse free rate of 95% • Overall survival rate of 99-100% • This rationale was to eradicate microscopic metastatic disease • However, many patients were unnecessarily radiated
Historically… • The use of surveillance for stage I NSGCT proved promising because of successful chemotherapy regimens for the relapse setting • This led to the investigation of surveillance for stage I seminoms
Surveillance • Rigshospitalet (Copenhagen) • Relapse rate of 17% at 5 years • Princess Margaret (Toronto) • Relapse rate of 15% at 5 years • Swedish Norwegian Testicular Cancer Study Group • Relapse rate of 14% with median relapse at 1.4 years • Salvage with Chemo (89%) or XRT (11%) with 1 failure • Chemo – 4 cycles cisplatin & etoposide • Primary site of relapse is in the paraaortic lymph nodes • Median time to relapse is 12-18 months though relapses after 6 years have been reported
Stage I Seminoma – Surveillance Predicting Recurrence • Warde et al, JCO 20:4448-4452, 2002 (pooled data from 4 major centers) • 5 year OS – 97.7% • 5 year CSS – 99.3% • 5 / 10 year RFS – 82.3% / 78.7%
Stage I Seminoma – Surveillance Predicting Recurrence • What factors predict recurrence? • Multivariate analysis found size >4cm (HR 2.0) and rete testis invasion (HR 1.7) predictive of relapse • 0 risk factors – 12.2% relapse • 1 risk factor – 15.9% relapse • 2 risk factors – 31.5% relapse Rete Testis 86% vs 77% SIZE 4cm 87% vs. 76%
Surveillance Schedules • ESMO (European Society for Medical Oncology) • 7 CT Scans of the abdomen over 5 years • NCCN (National Comprehensive Cancer Network) • 18 CT Scans of the abdomen/pelvis over 10 years • MRC randomized trial for surveillance (CT or MRI) • 3 in 3 years • 7 in 5 years
Stage I Seminoma - Surveillance • Surveillance – A reasonable option for stage 1 seminoma in patients who are committed to follow-up. (Warde et al JCO 13:2255, 1995, Von Der Maase et al, Eur Journ Cancer 1993;29A) • Follow up CT scans every 2-6 months for 2-3 years then less frequently • Pooled 5 year relapse rates of 18% • 84% of relapses occurred in the para-aortic nodes
Stage I Seminoma - Radiotherapy • In the modern era of post-orchiectomy radiation of the paraaortic lymph nodes… • OS rates are 92-99% • Cause-specific survival is nearly 100% • Relapse rates are 0.5-5% in modern studies (mostly supradiaphragmatic) • Most relapses occur <2 years from treatment (median 18 mo. in PMH study) • Chemotherapy is readily used in the setting of relapse
Stage I Seminoma – Radiotherapy Techniques • AP/PA field to the paraaortic and retroperitoneal lymph nodes +/- ipsalateral pelvic nodes • T10/11 superiorly to inguinal ligament inferiorly • Left renal hilum should be included for left sided tumors • Use CT to define volumes and avoid renal parenchyma • Scrotal irradiation is not required even in patients with scrotal violation
Stage I Seminoma – What Volume is Necessary • Due to low rate of pelvic node failure… • Phase II studies investigated paraaortic/RP nodal irradiation without pelvic irradiation • Showed very low rates of pelvic failure • Advantage of omitting pelvic radiation… • Decreased integral dose to patient • Decreased dose to the intact testicle!
Stage I Seminoma – Radiotherapy Volume • MRC Testicular Study Group (UK) • Fossa et al, JCO 17:1146-1154. 1999
Radiotherapy: 30 Gy/15 fx • upper border: disc - T10/T11 • lower border: mid-obturator foramen • ipsilateral margin: renal hilum to L5-S1, lateral edge of acetabulum • contralateral margin: transverse process to L5-S1, parallel to ipsilateral border, median border of obturator foramen • PA Field Identical above L5-S1
Stage I Seminoma – Radiotherapy Volume • Results • F/U 4.5 years • RFS was 96% in both arms • 4 pelvic failures – all in PA arm • 1 death from seminoma
Stage I Seminoma – Radiotherapy Volume • Paraaortic field resulted in improved • Diarrhea 7% vs 14% (p=0.013) • Leukopenia (p<0.00001) • Grade I – 14% vs 29% • Grade II – 5% vs 12% • Grade III Nausea/Emesis(p=0.08) • Time to normal sperm count improved with PA field despite more testicular shielding in DL group
Stage I Seminoma – Radiotherapy Dose • Jones et al, JCO, 23:1200-1208, 2005 • MRC/EORTC randomized 625 patients with para-aortic RT (DL if prior pelvic/scrotal surgery) to: • 30 Gy in 15 Fx, or • 20 Gy in 10 Fx • 30 Gy • 10 relapses (3 in field) • 20 Gy • 11 relapses (1 in field)
Stage I Seminoma – Radiotherapy Dose • Acute toxicity was worse with 30 Gy • Fatigue • Leukopenia • Nausea (p=0.06)
Stage I Seminoma – Chemotherapy • Oliver et al investigated the use of Carboplatin without radiation • 78 patients • 53 with 2 courses of Carbo • 25 with 1 course of Carbo • 44 months of follow up with only 1 relapse
The MRC (Oliver et at JCO, 29:957-962, 2011) randomized: With a median of 6.5 years follow up Relapse rate was 5.3% with carboplatin vs 4.0% with RT Stage I Seminoma – Chemotherapy 885 patients got PA or DL RT to between 20 and 30 Gy 560 patients got one injection of carboplatin
Stage I Seminoma – Chemotherapy • Improved fatigue with Carboplatin vs XRT • Fewer second primary GCT with Carboplatin (0.54%) vs XRT (1.96%) (p=0.04) • Paraaortic failures were the most common site with Carboplatin so surveillance imaging is required
Stage I Seminoma – Chemotherapy • Oliver et al (ASCO 2005 Abstract) • Pooled analysis of Phase II studies to compare 1 cycle vs. 2 cycles Carboplatin • Relapse Rate • 1 cycle – 4.4% • 2 cycles – 2.9% • No deaths due to seminoma
Stage I Seminoma - Outcomes • Combining the 3 randomized trials (2,466 patients) • 5 seminoma deaths for CSS of 99.8% • 4 relapses after 3 years • A prospective study of 1,192 patients treated in Sweden & Norway from 2000-2006 • 1 seminoma death for CSS of 99.9%
Stage I SeminomaSummary • Treatment • Inguinal Orchiectomy • Active Surveillance with serial imaging • ~85% RFS • 70% relapses <2years, nearly all <5 years • XRT • 95% RFS • Paraaortic equivalent with less toxicity than PA/Pelvic (dog leg) • 20 Gy equivalent with less toxicity than 30 Gy • Carboplatin • 95% RFS • One cycle equivalent to two cycles
Stage II Seminoma • Stage IIA – RP node <2cm • Stage IIB – RP node 2.1-5cm • Stage IIC - >5 cm • Few patients have stage II disease making randomized trials difficult to perform • Data hence stems from institutional experiences • The greatest prognostic factor is bulk of nodal disease (diameter of largest node)
Stage II Seminoma • NCCN guidelines recommend EBRT to RT to the para-aortics and ipsilateraliliacs (dogleg) to 35-40 Gy • PMH has reported on 95 pts treated in this way between 1981-1999 • 91% RFS with <5cm node • 44% RFS with >5cm node • Relapse most common in mediastinum, SCF, lung, bone • 70% salvaged with chemotherapy
Residual Retroperitoneal Mass • Presence of residual masses after definitive treatment is common • Most often represent fibrosis or necrosis • Very few contain viable tumor • Options • Observation • Surgery • RT (after chemo) • PET is of little value in this setting
Residual Retroperitoneal Mass • MSKCC studies patients with residual RP masses and the role of surgery or biopsy • 0% of patients with mass <3cm had viable tumor • These patients should be observed • 30% of patients with mass >3cm had viable tumor • Biopsy residual mass or… • Observe with serial CT • Treat with enlarging mass
Residual Retroperitoneal Mass • Horwich et al reported 29 patients with residual RP mass following platinum-based chemotherapy • 15 treated with adjuvant XRT • 14 observed • No evidence that adjuvant XRT decreased relapse rate • MRC pooled 123 patients residual mass • 56% treated with adjuvant XRT • 44% observed • No evidence that adjuvant XRT decreased relapse rate
Stage II Seminoma • XRT yields RFS of 90% with PA/Pelvis fields • Platinum based chemotherapy can be used with adjuvant XRT showing no benefit • Residual RP mass • <3cm - nearly always fibrotic – observe • >3cm – 30% residual tumor – biopsy or serial CT
Stage IIC - III Seminoma • These patients are uncommon (5%) • 40-50% relapse rate for IIC patients treated with EBRT alone • 4 cycles of Cisplatin/Etoposide has yielded 92% freedom from progression at MSKCC and is considered the standard approach
Targeted Therapy • Scientists evaluating cell surface molecules and mutations associated with GCT • No targeted therapy in clinical use • With high RFS, CSS, OS rates, targeted therapy is not likely to be beneficial or cost effective
Proton Therapy • Dosimetric models suggest that there is decreased normal tissue exposure with protons • No patients being treated with protons • Not cost effective
Fertility • 50 percent of men with testicular cancer have some impairment of spermatogenesis (more with NSGCT than seminoma) • All patients should have sperm analysis with option of cryopreservation prior to adjuvant therapy • RT patients should have shielding of the contralateral testicle
Fertility & Radiation • Temporary azospermia can occur with 2 Gy • Permanent azospermia after ~7 Gy • Stem cells are the most sensitive to XRT resulting in azospermia • Leydig cells (testosterone producing cells) have a much higher tolerance so patients rarely suffer from hypotestosterone from XRT
Fertility • Norwegian study of 1,814 patients • 554 attempted natural conception • Equal rates of conception with RT compared to retroperitoneal LN dissection (~80%)