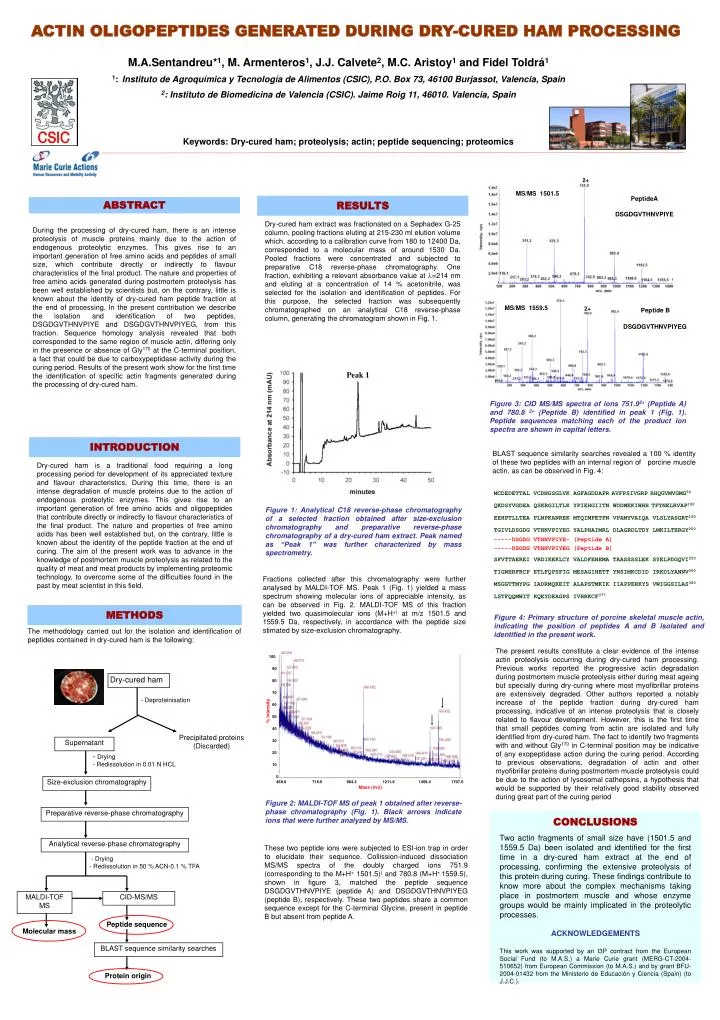
ACTIN OLIGOPEPTIDES GENERATED DURING DRY-CURED HAM PROCESSING
E N D
Presentation Transcript
2+ MS/MS 1501.5 PeptideA DSGDGVTHNVPIYE MS/MS 1559.5 2+ Peptide B DSGDGVTHNVPIYEG METHODS The methodology carried out for the isolation and identification of peptides contained in dry-cured ham is the following: 100 90 Dry-cured ham 80 70 - Deproteinisation 60 % Intensity 50 40 Precipitated proteins (Discarded) Supernatant 30 20 - Drying - Redissolution in 0.01 N HCL 10 0 Size-exclusion chromatography 469.0 716.6 964.2 1211.8 1459.4 1707.0 Mass (m/z) Preparative reverse-phase chromatography Analytical reverse-phase chromatography - Drying - Redissolution in 50 % ACN-0.1 % TFA MALDI-TOF MS CID-MS/MS Peptide sequence Molecular mass BLAST sequence similarity searches Protein origin M.A.Sentandreu*1, M. Armenteros1, J.J. Calvete2, M.C. Aristoy1and Fidel Toldrá1 1:Instituto de Agroquímica y Tecnología de Alimentos (CSIC), P.O. Box 73, 46100 Burjassot, Valencia, Spain 2: Instituto de Biomedicina de Valencia (CSIC). Jaime Roig 11, 46010. Valencia, Spain Keywords: Dry-cured ham; proteolysis; actin; peptide sequencing; proteomics ACTIN OLIGOPEPTIDES GENERATED DURING DRY-CURED HAM PROCESSING Peak 1 Figure 3: CID MS/MS spectra of ions 751.92+ (Peptide A) and 780.8 2+ (Peptide B) identified in peak 1 (Fig. 1). Peptide sequences matching each of the product ion spectra are shown in capital letters. MCDEDETTAL VCDNGSGLVK AGFAGDDAPRAVFPSIVGRPRHQGVMVGMG50 QKDSYVGDEAQSKRGILTLK YPIEHGIITN WDDMEKIWHHTFYNELRVAP100 EEHPTLLTEA PLNPKANREKMTQIMFETFN VPAMYVAIQA VLSLYASGRT150 TGIVLDSGDG VTHNVPIYEG YALPHAIMRLDLAGRDLTDY LMKILTERGY200 -----DSGDG VTHNVPIYE- (Peptide A) -----DSGDG VTHNVPIYEG (Peptide B) SFVTTAEREIVRDIKEKLCY VALDFENEMA TAASSSSLEKSYELPDGQVI250 TIGNERFRCP ETLFQPSFIGMESAGIHETT YNSIMKCDID IRKDLYANNV300 MSGGTTMYPG IADRMQKEIT ALAPSTMKIKIIAPPERKYS VWIGGSILAS350 LSTFQQMWITKQEYDEAGPS IVHRKCF377 Figure 4:Primary structure of porcine skeletal muscle actin, indicating the position of peptides A and B isolated and identified in the present work. Figure 1: Analytical C18 reverse-phase chromatography of a selected fraction obtained after size-exclusion chromatography and preparative reverse-phase chromatography of a dry-cured ham extract. Peak named as “Peak 1” was further characterized by mass spectrometry. Figure 2: MALDI-TOF MS of peak 1 obtained after reverse-phase chromatography (Fig. 1). Black arrows indicate ions that were further analyzed by MS/MS. CONCLUSIONS Two actin fragments of small size have (1501.5 and 1559.5 Da) been isolated and identified for the first time in a dry-cured ham extract at the end of processing, confirming the extensive proteolysis of this protein during curing. These findings contribute to know more about the complex mechanisms taking place in postmortem muscle and whose enzyme groups would be mainly implicated in the proteolytic processes. ACKNOWLEDGEMENTS This work was supported by an I3P contract from the European Social Fund (to M.A.S.) a Marie Curie grant (MERG-CT-2004-510652) from European Commission (to M.A.S.) and by grant BFU-2004-01432 from the Ministerio de Educación y Ciencia (Spain) (to J.J.C.).