Understanding Infrared Spectroscopy: Principles and Applications in Organic Chemistry
This document provides a comprehensive overview of infrared (IR) spectroscopy and its significance in organic chemistry. It explores the relationship between radiant energy, frequency, and wavelength, and details various types of electromagnetic radiation. The content specifically focuses on the application of IR spectroscopy for structural analysis of organic molecules, discussing characteristic absorption ranges for functional groups, interpreting IR spectra, and identifying key molecular vibrations. This knowledge aids chemists in confirming the presence of functional groups within compounds.

Understanding Infrared Spectroscopy: Principles and Applications in Organic Chemistry
E N D
Presentation Transcript
Spectroscopy: IR Renee Y. Becker Valencia Community College CHM 2011
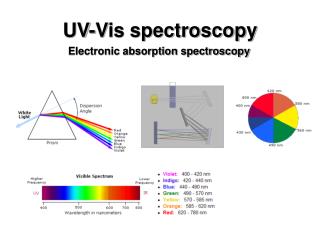
Radiant energy is proportional to its frequency (cycles/s = Hz) as a wave (Amplitude is its height) • Different types are classified by frequency or wavelength ranges
γ-rays (gamma rays)- greatest energy and highest frequency. Emitted from some radioactive nuclei. Can cause biological damage. • X-rays- lower in energy than γ -rays. Can cause biological damage in high doses. • Ultraviolet (UV) light- can cause sunburn and even skin cancer. • Visible light- speaks for itself ! • Infrared radiation (IR) - heat! • Microwaves- we cook with them and also used in radar. • Radio waves- lowest frequency. Radio and TV transmissions and NMR spectroscopy.
The mode of propagation of electromagnetic radiation is the wave. • The wave is characterized by its wavelength (), frequency (), and amplitude. • Frequency () units: s-1 or hertz (Hz) • The intensity of a wave is proportional to the square of its amplitude. • Electromagnetic radiation travels at constant velocity in a vacuum: 3.00 x 1010 cm/s (speed of light).
Wavelength x Frequency = Speed • (cm) x (s-1) = c (cm/s)
Thanks to Max Planck and Al Einstein: E = Energy of 1 photon (1 quantum) h = Planck’s constant (6.62 x 10-34 J·s = 1.58 x 10-34cal·s) = Frequency (s-1) = Wavelength (cm) c = Speed of light (3.00 x 1010 cm/s)
The energy of a photon varies directly with the frequency and inversely with the wavelength • High frequencies and short wavelengths = higher energies • Low frequencies and long wavelengths = lower energies
Infrared Spectroscopy of Organic Molecules • IR region lower energy than visible light (below red – produces heating as with a heat lamp) • 2.5 x 10-4 cm to 2.5 x 10-3 cmregion used by organic chemists for structural analysis • IR energy in a spectrum is usually measured as wavenumber (cm-1), the inverse of wavelength and proportional to frequency
Specific IR absorbed by organic molecule related to its structure • The IR spectrum covers the range from 4000 cm-1 to 400 cm-1 • This represents energy ranges from 48.0 - 4.80 kJ/mol (11.5 - 1.15 kcal/mol).
IR energy absorption corresponds to specific modes, corresponding to combinations of atomic movements, such as bending and stretching of bonds between groups of atoms called “normal modes” • Energy is characteristic of the atoms in the group and their bonding • Corresponds to vibrations and rotations
Interpreting Infrared Spectra • Most functional groups absorb at about the same energy and intensity independent of the molecule they are in • Characteristic higher energy IR absorptions in Table 12.1 can be used to confirm the existence of the presence of a functional group in a molecule • IR spectrum has lower energy region characteristic of molecule as a whole (“fingerprint” region; 1300 to 625 cm-1) • See samples in Figure 12-13
4000-2500 cm-1 N-H, C-H, O-H (stretching) 3300-3600 N-H, O-H 3000 C-H 2500-2000 cm-1 CºC and C º N (stretching) 2000-1500 cm-1 double bonds (stretching) C=O 1680-1750 C=C 1640-1680 cm-1 Below 1500 cm-1 “fingerprint” region Regions of the Infrared Spectrum
Differences in Infrared Absorptions • Molecules vibrate and rotate in normal modes, which are combinations of motions (relates to force constants) • Bond stretching dominates higher energy modes • Light objects connected to heavy objects vibrate fastest: C-H, N-H, O-H • For two heavy atoms, stronger bond requires more energy: C º C, C º N > C=C, C=O, C=N > C-C, C-O, C-N, C-halogen
Infrared Spectra of Hydrocarbons • C-H, C-C, C=C, C º C have characteristic peaks • absence helps rule out C=C or C º C
IR: Alcohols and Amines • O–H 3400 to 3650 cm1 • Usually broad and intense • N–H 3300 to 3500 cm1 • Sharper and less intense than an O–H
IR: Aromatic Compounds • Weak C–H stretch at 3030 cm1 • Weak absorptions 1660 - 2000 cm1 range • Medium-intensity absorptions 1450 to 1600 cm1 • See spectrum of phenylacetylene, Figure 12.15
IR: Carbonyl Compounds • Strong, sharp C=O peak 1670 to 1780 cm1 • Exact absorption characteristic of type of carbonyl compound • 1730 cm1 in saturated aldehydes • 1705 cm1 in aldehydes next to double bond or aromatic ring
C=O in Ketones • 1715 cm1 in six-membered ring and acyclic ketones • 1750 cm1 in 5-membered ring ketones • 1690 cm1 in ketones next to a double bond or an aromatic ring
C=O in Esters • 1735 cm1 in saturated esters • 1715 cm1 in esters next to aromatic ring or a double bond
Identify the functional groups in compounds that are responsible for the following absorptions: • A compound with a strong absorption at 1710 cm-1 • A compound with a strong absorption at 1540 cm-1 • A compound with a strong absorption at 1720 cm-1 and at 2500-3100 cm-1
How might you use IR spectroscopy to distinguish between the following pairs of isomers? • CH3CH2OH and CH3OCH3 • Cyclohexane and 1- hexene