Understanding Phases and Phase Changes: Solids, Liquids, Gases, and Plasma
This overview explores the different phases of matter: solid, liquid, gas, and plasma, focusing on their properties and phase changes. Key phase changes such as freezing, melting, and boiling of water illustrate how temperature remains constant during these processes. For instance, water freezes at 0°C, remains at 0°C while freezing, and melts back to liquid at the same temperature. Similarly, water boils at 100°C, with temperature constant during boiling. Understanding these concepts is crucial for grasping the behavior of materials and energy transformations.

Understanding Phases and Phase Changes: Solids, Liquids, Gases, and Plasma
E N D
Presentation Transcript
The Phases Coldest Hottest Solid Liquid Gas Plasma
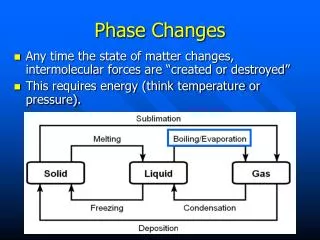
Energy during a Phase Change Example) Water Freezes (liquid to solid) at 0oC Stays constant at 0oC during freezing
Energy during a Phase Change Example) Water Melts (solid to liquid) at 0oC Stays constant at 0oC during melting
Energy during a Phase Change Example) Water Boils (liquid to gas) at 100oC Stays constant at 100oC during boiling
Phase Changes • In summary: During a phase change, the temperature is CONSTANT!!!