New Intercalation Materials
New Intercalation Materials. Grant DMR-0313963 PI: M. Stanley Whittingham. Objectives: Synthesize new host structures for intercalation reactions and identify the key parameters for new structure formation and optimum reactivity.

New Intercalation Materials
E N D
Presentation Transcript
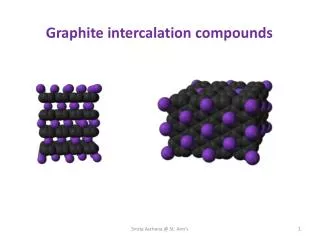
New Intercalation Materials Grant DMR-0313963 PI: M. Stanley Whittingham • Objectives: • Synthesize new host structures for intercalation reactions and identify the key parameters for new structure formation and optimum reactivity. • Elucidate their chemical and physical properties, including stability, structure, electrochemistry and electronic/magnetic behavior. • Achievements: • The structures of several new vanadium oxides and phosphates have been determined, and the controlling parameters identified. • The use of mild hydrothermal synthesis and electro-deintercalation have been successfully employed in the synthesis of some oxides and phosphates. • An understanding of the electrochemistry is being developed, and the basics of the complex magnetic behavior of the planar and scrolled vanadium oxides, as well as of the mixed Li(MnCoNi)dioxides are in hand. (e) Schematic of (a) layered vanadium oxides bending (b) in the hydrothermal reaction of vanadium oxides to form (c) nanoscrolls and (d) nanotubes. (Phys. Rev. B69: 085410, 2004). (e) shows the vanadium oxide nanotubes.
New Intercalation Materials Grant DMR-0313963 PI: M. Stanley Whittingham This project targets the discovery and characterization of new transition metal oxides that are capable of acting as the host for guest-host reactions - intercalation compounds. Little is still understood about the relationship between the crystal structure of materials and their chemical and physical properties. We have begun to get a better understanding of what controls the reactions of inorganic materials, so that we can predict what compounds will be formed. However, the inorganic/materials chemist is still far behind the synthetic organic chemist who can map out a synthetic sequence. One of our goals is to further the understanding of synthetic approaches, and our strategy is to use ‘soft chemistry’ conditions where it is possible to control the building blocks that eventually are used for building the structure. The vanadium oxides are particularly suitable for this study, because many structural types can be formed with close compositions. Thus the nature of the impact of the building block bonding can be readily studied. The example on this slide shows the bending of sheets of vanadium oxide to form nanoscrolls or tubes. What impact does this have on the properties of the material, and why does this bending occur? The compounds readily undergo ion exchange but not with lithium; however they very readily react under redox reactions with lithium, but these reactions are strangely strongly dependent on the electrolyte salt for the tubes unlike the flat starting materials. The structures were elucidated in collaboration with a group at Michigan Technological University, and the optical behavior with J. Musfeldt at U. Tennessee. The bonding must be strained as the sheets are curved and we are trying to control the degree of bending to see if and how the properties change. N. Chernova (postdoc at Binghamton) is determining their magnetic behavior as a function of guest specie and degree of reduction - she is finding the behavior is complex with a mix of dimers and isolated spins. Lithium reduction (harsh with butyl lithium and mild with lithium iodide) does change the magnetic behavior, but in no case is ferromagnetic behavior at room temperature found (as reported by researchers at IBM). Understanding their behavior is critical to potential uses as in battery electrodes, heterogeneous catalysis, electromechanical devices or in optical systems. If the degree of curling can be controlled in-situ by a simple redox reaction, one might envision a range of potential applications.
c b a New Intercalation Materials Grant DMR-0313963 PI: M. Stanley Whittingham • Education and Outreach: • Graduate and undergraduate students, postdocs and visitors. Graduated PhD students now in industry. • Courses in academia and lectures to industry. • Results applied to US energy needs (DOE). • Knowledge applied to two battery and IP litigations. • Collaborations and Applications: • The interdisciplinary nature of this work has demanded collaborations with other academics and with the National Laboratories. These have successfully solved several problems. • Several of the materials synthesized are now being evaluated as cathode materials in lithium batteries for hybrid electric vehicles (DOE et al). • Hydrothermal synthesis is under consideration for the large scale and low cost processing of phosphates (DOE, and SBIR). (a) (Top) The structure (a) and electrochemistry (b) of a 3d iron phosphate showing the lithium ions (spheres) (Chem. Mater. 17, 1139, 2005). (Bottom) The layered structure of Li(MnNiCo)O2, and it’s magnetization, indicating the presence of nickel ions in the lithium layer at certain compositions (Chem. Rev.104, 4271, 2004).
New Intercalation Materials Grant DMR-0313963 PI: M. Stanley Whittingham The project continues to provide educated scientists for the United States, with many of the students going to US industry (sometimes after with a postdoc). A few also move on to educate the next generation of scientists. A number of new courses have been developed, which not only include the traditional technical content but now also include a high level of communications competence. This latter was noted as a major weakness by US industry in PhD graduates. In addition a new graduate program in Materials Science was initiated and the first MS and PhD students graduated this summer - students now have the option of either the more fundamental Chemistry degree of the more applied Materials degree. The complex interdisciplinary nature of our research demands that we have collaborators. In addition to those mentioned previously we are working with Clare Grey from SUNY Stony Brook to understand the atomic structure of the iron phosphate shown - where are the protons and the lithium; with Brookhaven using the synchroton on the structures of many compounds including the layered oxide as a function of lithium content, and with NIST on the ordering of the nickel ions. The most promising materials are being investigated with Berkeley National Lab. (DOE funding) for possible use in lithium batteries for HEVs. There is much interest in the potential use of hydrothermal synthesis for the formation of materials on the commercial level - our lab is perceived to be the leading proponent in the US. The iron phosphate pictured here was formed hydrothermally, and the optimum electrochemical behavior was obtained after an ion-exchange treatment to replace many of the protons with lithium ions. We have made the commercially interesting olivine iron phosphate hydrothermally, but there are some issues with the lithium and iron ordering that we are now studying. We have been approached as to whether the layered oxide (pictured in the lower figure) can be made hydrothermally, and will be exploring that but ion ordering may well be a problem again. The magnetization behavior shown here is a strong indicator of the presence of nickel ions in the lithium layer. The presence of nickel allows interactions between the ions in adjoining transition metal layers which leads to the hysteresis in the magnetization. No nickel ions in the lithium layer = no interaction between the transition metal layers = no hysteresis. Surprisingly we found that a certain level of nickel is a good pinner of the layers resulting in greater structural retention when all the lithium is removed. This NSF funded program is leading to a better understanding of the fundamental behavior of these oxides and phosphates, which is being used for their battery applications. This program is also providing well-educated scientists to US industry, and the demand is higher than we can supply. The knowledge is also being requested as expertise in some court cases.