Chemical Equilibrium
This overview explains the concepts of chemical equilibrium, including static and dynamic equilibrium. It delves into Le Chatelier’s Principle, demonstrating how systems at equilibrium react to changes in temperature, pressure, and concentration. By analyzing examples such as the formation of ammonia in the Haber process, the interplay between forward and reverse reactions is explored. The description emphasizes practical applications in industries like fertilizer production, highlighting optimal conditions for maximizing yield.

Chemical Equilibrium
E N D
Presentation Transcript
Static Equilibrum The entire system is not moving Eg: A meter stick that is suspended at its centre pf gravity. Dynamic Equilibrum The system is in motion. Two opposing motions balance each other out. Eg: a man on a threadmill. Chemical Equilibrium
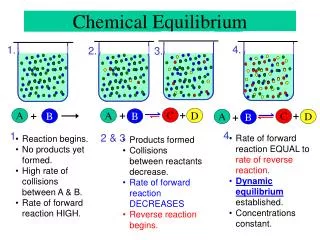
Some reactions go to completion. Eg: Magnesium burning in air. 2Mg + O2→ 2MgO Many chemical reactions don’t go to completion (equilibrum reactions). Take place in both directions (reversable) Eg: formation of ammonia from H2 + N2 3H2 + N2 2NH3 Dynamic equilibrium Chemical Equilibrium
Chemical Equilibrium • At equilibrum the rate of the forward reaction equals the rate of the reverse reaction. • The conc at equilibrum remains constant
Chemical Equilibrium • Definition: Chemical Equilibrum is a state of dynamic balance where the rate of the forward reaction equals the rate of the reverse reaction.
Le Chatelier’s Principle • 1888 Henri Le Chatelier put forward a rule which allowed chemists to predict the direction taken by an equilibrum rxn when the conditions of the rxn were changed. • RULE:If a stress is applied to a system at equilibrum, the system readjusts to relieve the stress applied. • Stress = change in condition of the reaction eg temp/conc/pressure • Catalyst=reaches equili quicker
Le Chatelier’s Principle • When a stress is applied the system will react to minimise the stress • Ie: return to its original equilibrum
Le Chatelier’s Principle • Consider:
Each test tube is at equilibrum • What happens when palced in ice? • Light Yellow (more dinitrogen tetraoxide made) • What happens when placed in hot water? • Darker Brown (more nitrogen dioxide made)
What were the stresses? • The change in temperature hot and cold. • When placed in hot water it carried out the reaction which would absorb heat • When placed in cold water it carried out the reaction which would create heat • This is to minimise the effect of the stress
Effects of Changing Pressure of Gases in rxns • If pressure is increases the equilibrium is shifter the side of the equation which has the smaller number of molecules. • Why? • The smaller number of molecules will occupy less volume thus reduce the pressure
Le Chatelier’s Principle • State Le Chatlier’s Principle: • Le Chatlier’s Princilpe states that if a stress is applied to a system at equilibrum the system will readjust to relieve/minimise the applied stress
Le Chatelier’s Principle Predicts: • That in an all gaseous rxn an increase in pressure will favour the rxn which takes place with reduction in volume ie, towards the side with the smaller number of molecules • Only affects equili rxns with unequal no’s of gaseous reactants and products
Equilibrium and Catalysts • A catalyst speeds up the rate at which equilibrium is reached but does not change the position of equilibrium. • Increases both the rate of the forward and reverse reactions
Mandatory Experiment • To demonstrate Le Chatelier’s Principle: • A) Effect of • Temp • Conc changes
What happens when • Add HCl • Turns blue • Add water • Turns red • Place in hot water • Turns blue
To demonstrate Le Chatelier’s Principle: • B) Effect of Concentration • Originally the soln is orange. • Where does the equilibrium lie?
What happens when • Add dilute NaOH • Turns yellow • Add dilute HCl • Turns orange
To demonstrate Le Chatelier’s Principle: • C) Effect of Concentration • Originally the soln is red. • Where does the equilibrium lie?
What happens when • Add dilute HCl • Turns blue • Add iron(III) chloride • Turns red
Industrial Applications of Le Chatelier’s Principle • Manufacture of ammonia by the Haber Process • Manufacture of sulfuric acid by the contact process
The Haber Process • Use: Fertilisers (80%), explosives Cleaning agents • Objective: Produce as much max amount of NH3 as cheaply as possible. • What used: • H2 from Natural Gas • N2 from Air
The Haber Process • The amount of NH3 produced depends on: • Temperature • Pressure • Catalyst (Iron)
Achieving max yield Pressure: • Increased pressure at equilibrium favours the production of NH3 • Carried out at high pressure (200 atmospheres) • Pressure cant be too high: • Expensive to build high pressure plants • Problems like safety
Temperature: • A lower temp will cause more ammonia to be produced • Temp cant be too low: • Rate of reaction will be to slow • Not enough collisions having energy = activation energy • Temp used = 500°C
Catalyst: • In absence of catalyst the rxn has a high activation energy. • Catalyst used because: • Brings system to equilibrium faster by lowering Eact • Rxn can now proceed at a lower temp reducing fuel costs
Thus: • Le Chatelier’s Principle tells us that the best conditions to produce NH3 in the Haber process is under conditions of: • High pressure • Low temperature
Manufacture of sulfuric acid by the contact process • Manufactured by the Contact Process • H2SO4 used in the manufacture of paints,detergents, fertilisers, plastics, fibres,car batteries. • Sulfur trioxide is the desired product as it dissolves in water to give H2SO4.
The Contact Process • Called the contact process as very close contact must be made between the two reactant gases and the catalyst. • The amount of SO3 produced depends on: • Temperature • Pressure • Catalyst • Concentration
Temperature: • A lower temp will cause more SO3 to be produced • Temp cant be too low: • Rate of reaction will be to slow • Not enough collisions having energy = activation energy • Temp used = 450°C used as found the catalyst works best at this temp.
Pressure: • Increased pressure at equilibrium favours the production of NH3 • Carried out at a pressure just higher than atmospheric pressure. • Pressure cant be too high: • Expensive to build high pressure plants • Problems like safety
Concentration • By removing the SO3 as it is formed reduces its conc thus shifting the equilibrium to the right
Catalyst • Brings the reaction to equilibrium faster by reducing the activation energy.
The Equilibrium constant • Calculations to find the conc of products & reactants at equilibrm • System must be at equilibrium • Temperature must stay constant
The Equilibrium constant • Every equilibrum rxn has its own value for Kc at a particular temperature • The larger the value for Kc the futher the equili is pushed towards the products. • The smaller the value for Kc the futher the equili is pushed towards the reactants. • We do not need to know the unit of Kc.