Limestone
Limestone. Limestone .

Limestone
E N D
Presentation Transcript
Limestone • Limestone is made of shells and dead sea creatures that collect at the bottom of the ocean. They get buried by other sediment and, after millions of years of being under pressure, the particles get cemented together to form solid rock. Limestone is a very strong rock and is often cut into large blocks and used in the construction of buildings and skyscrapers. Crushed limestone is also used to make cement, plaster and chalk. Limestone fizzes in vinegar, which means that it reacts to acids. This also means that the outsides of many of our buildings and monuments are being affected by acid rain. Acid rain occurs when large companies pollute the air by releasing chemicals like sulfur into the atmosphere. The sulfur combines with hydrogen and oxygen in the air to form sulfuric acid.
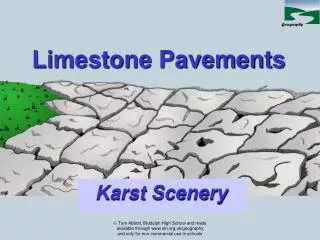
Limestone is also Sedimentary rock composed mainly of calcium carbonate, usually in the form of calcite and, less commonly, aragonite. It may contain considerable amounts of magnesium carbonate (dolomite) as well. Most lime-stones have a granular texture; in many cases, the grains are tiny fragments of fossil animal shells. Much knowledge of the Earth's history has been derived from the study of fossils embedded in limestone and other carbonate rocks. Limestone is used as a soil conditioner, in the manufacture of glass, and in agriculture. Ornamental varieties are used for flooring, exterior and interior facings of buildings, and monuments. This is limestone pavement
What is limestone used for? Limestone has many uses. Those uses include patio pavers, landscaping rock, road gravel, concrete aggregate and soil conditioner. In addition, limestone is used as an additive to paint and other products, and a source of calcium carbonate which is used in a great deal of products including antacids. Cooked limestone is used to make Portland cement, the key ingredient in mortars, cements, and concrete. Here are some things that limestone makes Glass making Ingredient in toothpaste Bread and cereals as a source of calcium Making paper white Sugar purification Making brake pads Preparation of wools and dyes Climbing walls A suppression of methane explosions in underground coalmines Manufacturing of quicklime (calcium oxide) and slaked lime (calcium hydroxide). Manufacturing of steel Lower water pH Food preservation
Thank you for watching Matt, Jamie, Ollie and Sam