NUCLEOTIDE BIOSYNTHESIS
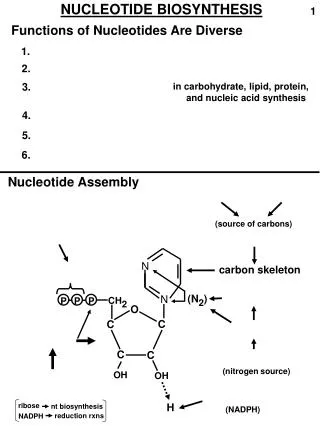
NUCLEOTIDE BIOSYNTHESIS. N. N. O. C. C. C. C. P. P. P. ribose. nt biosynthesis. reduction rxns. NADPH. 1. Functions of Nucleotides Are Diverse. 1. 2. 3. in carbohydrate, lipid, protein, and nucleic acid synthesis. 4. 5. 6. Nucleotide Assembly. (source of carbons).

NUCLEOTIDE BIOSYNTHESIS
E N D
Presentation Transcript
NUCLEOTIDE BIOSYNTHESIS N N O C C C C P P P ribose nt biosynthesis reduction rxns NADPH 1 Functions of Nucleotides Are Diverse 1. 2. 3. in carbohydrate, lipid, protein, and nucleic acid synthesis 4. 5. 6. Nucleotide Assembly (source of carbons) carbon skeleton (N2) CH2 (nitrogen source) OH OH H (NADPH)
CH2 H CH2 NH2 H H H H H H H O Activation of the Ribose Sugar for Nt Biosynthesis OH OH OH OH 2nd step H CH2 O O O O H CH2 C C C C C C C C H H H O H H C C C C C C C C H OH OH OH P P P P P P P P OH OH Nucleotide Synthesis 2 activated ribose (PRPP) + ATP + amino acids (CO2 + N2) activated ribose (PRPP) + base 1st step sugar ring is activated at the a-configuration 5-phospho-ribosyl- 1-pyrophosphate ribose-5-PO4 N9 of base
3 CH2 NH2 H H H H OH OH R5P PRPP O 5-phopho-b-ribosyl amine C C (ring assembly) C C inosine monophosphate GMP AMP P ADP GDP ATP GTP N9 of Pu base amine is now attached in the b configuration this becomes the b-glycosidic bond PRPP amidotransferase inhibitors of PRPP amidotransferse
4 Purine Nucleotide Biosynthesis H purine ring N N N 2HN N H H O Source of Purine Ring Constituents N 6 7 5 N 1 8 4 2 9 N 3 N R N CH2 10-formyl-tetrahydrofolate
PURINE PATHWAY NH2 CH2 H CH2 H H H H H H H O OH OH OH OH CH2 H H H N H H CH2 CH OH OH O C NH O O O H O O ribose-5-phosphate (R5P) C C C C C C N CH2 CH C C C C C C O HN C P P P P P NH R5P 6 (5-phospho-ribosyl- 1-pyrophosphate) GLN + H2O GLU + PPi amidophosphoribosyl transferase NH2 formylglycinamide ribotide (FGAR) GAR synthetase CH2 ADP + Pi C NH GAR transformylase R5P THF N10-formyl-THF (tetrahydrofolates are methyl/formyl donors) glycinamide ribotide (GAR) FGAM synthetase ATP + GLN + H20 ATP ADP + Pi ADP + GLU + Pi AIR synthetase
7 N N C HC 5 member imidazole ring CH CH C H2N H2N H2N H2N C N N R5P R5P R5P R5P R5P ATP + HCO3 N O O O O C C CH H H C C NH NH C C N O CH2 CH2 O O C C O O N C C H2N CH C N CH N H N C H C H2N C CH O CH C C C C O O O N O C N10-formyl-THF (formyl donor) O O O O 5-aminoimidazole ribotide (AIR) carboxyaminoimidazole ribotide AIR carboxylase now have 4 members of the 5 member ring ADP + Pi SACAIR synthetase 5-aminoimidazole-4- (N-succinylocarboxamide) ribotide (SACAIR) ATP ADP + Pi 5-formaminoimidazole- 4-carboxamide ribotide (FAICAR) 5 of the 6 ring members adenylosuccinate lyase all 6 ring members THF 5-aminoimidazole-4- carboxamide ribotide (AICAR) AICAR transformylase fumarate
8 N C HN IMP cyclohydrolase CH C HC 4 N 5 3 H2O 6 2 N 1 NH2 OH O CH2 C O N C O C NH2 H O H H H CH2 H O C O OH OH OH two components joined ring is closed PO3 O C N H O C C C C P IMP is the precursor for purine exocyclic nt modifications aspartate is the N donor glutamine is the N donor Pyrimidine Synthesis Pyrimidine ring assembled attachment to the ribose sugar (precursor of UMP)
GLU carbamyl phosphate 2 ATP + HCO3 + GLN + H2O 9 The synthesis of carbamyl phosphate and regulation of pyrimidine biosynthesis carbamyl phosphate synthetase II PRPP and IMP (from PU synthesis) pyrimidine nucleotides Why?? Why not regulate Pu and Pyr synthesis at PRPP synthetase?? 5-phospho-b-ribosyl amine R5P PRPP PRPP synthetase PRPP amido transferase carbamyl PO4 synthetase II Pu nts Pyr nts inhibits inhibits
10 Pyrimidine Biosynthesis
PYRIMIDINE PATHWAY A A O O CH2 OH OH OH OH CH2 P P P P P P P P NH2 O H HO C C CH2 NH3 HO C CH2 CH2 C CH2 H C O O NH2 HO H H CH C C O C C C C C C H2N CH2 CH2 O O O O N O C O NH3 NH3 H O O O O O O O O 11 start with 4 substrates (building the Pyr ring starting with carbonate) + 1 2 H2O (ATP supplies energy & phosphate) carbamoyl phosphate synthetase 1 + 2 aspartate transcarbamylase constituents of Pyr are now joined H2O (dehydration) dihydroorotase
12 C CH2 HN CH N H C CH HN C C H CH2 N CH HN H C H H H O N C OH OH CH HN CH O O O N CH2 C C C C C C C C C C C C C O O O C C C O O O O H C C C H H O O O O H O O O P P P P P OH OH reduced quinone quinone double bond formation dihydroorotate dehydrogenase (5-phospho-ribosyl- 1-pyrophosphate) a-configuration orotate phosphoribosyl transferase in contrast to the purines, the base is synthesized before attachment to ribose CH2 b configuration OMP decarboxylase H H H H CO2 OH OH
XDP XTP XMP CH2 CH2 base base O O H H C C C C H H H H H H C C C C OH OH OH H P P P P 13 two points concerning nucleotide biosynthesis 1. 2. diphosphate / triphosphate synthesis source of ATP deoxyribonucleotide synthesis ubiquitous enzyme found in all organisms substrate ribonucleotide reductase ribonucleotide reductase XDPs
14 NADPH thioredoxin ribonucleotide reductase reduced reduced NADP thioredoxin ribonucleotide reductase oxidized oxidized reduced oxidized a1 a2 b 2 b 1 ribonucleotide reductase “redox” rxn (like cytochromes in oxidative phosphorylation) ribonucleotide reductase recycled through a series of oxidation - reduction steps/rxns ribonucleotide reductase (E. coli) ( XTP / XDP / XMP binding sites ) ( inhibitors and activators of Pu and Pyr pathways )