Condensation polymers
Condensation polymers. C.8.1 Distinguish between addition and condensation polymers in terms of their structures. C.8.2 Describe how condensation polymers are formed from their monomers. C.8.3 Describe and explain how the properties of polymers depend on their structural features.

Condensation polymers
E N D
Presentation Transcript
Condensation polymers C.8.1 Distinguish between addition and condensation polymers in terms of their structures. C.8.2 Describe how condensation polymers are formed from their monomers. C.8.3 Describe and explain how the properties of polymers depend on their structural features. C.8.4 Describe ways of modifying the properties of polymers. C.8.5 Discuss the advantages and disadvantages of polymer use.
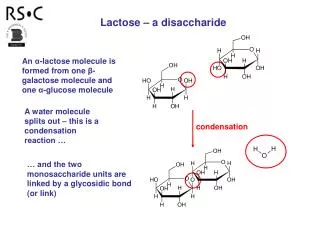
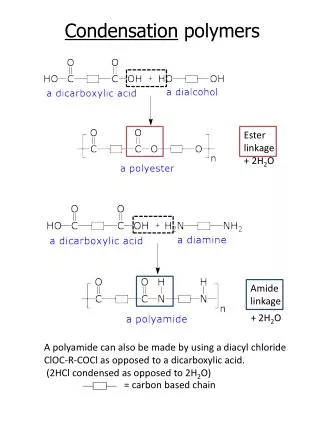
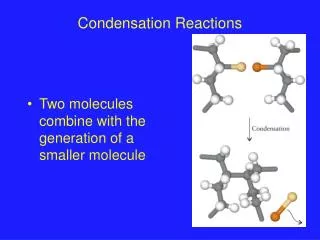
Addition polymer formed by an addition reaction, where many monomers bond together via rearrangement of bonds without the loss of any atom or molecule. Usually made from alkenes Condensation polymer formed by a condensation reaction where a molecule, usually water, is lost during the formation Made from monomers that contain at least two reactive functional groups( or same functional group at least twice) Distinguish between addition polymer and condensation polymer
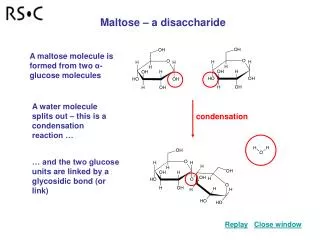
Formation of polyester • Polyethen terephthalate (PET) • When benzene-1,4-dicarboxylic acid and ethane-1,2 diol reacts
Phenol-methanal plastic • The reaction is a condensation polymerisation in which water is eliminated as a hydrogen atom from the benzene ring of each of two neighbouring phenol molecules combines with the oxygen atom from a methanal molecule ( When acid or alkali is added to the mixture of phenol and methanal) • The remaining –CH2– group from the methanal molecule then forms a bridge between two neighbouring phenol molecules. This process, repeated many thousands of times, forms chains of phenol and methanal molecules linked in this way.
Mechanism • When acid or alkali is added to the mixture of phenol and methanol. • Methanal is substituted in the 2- or 4- position in the phenol • It then undergoes a condensation reaction with another phenol molecule.
Long Chain • Long chain of phenol-methanal plastics (covalent cross linking – three dimensional structure) is built up when further polymerization takes place
Formation of Polyurethane • When polyhydric alcohols (e.g. diols or triols) reacts with compounds containing more than one isocyanate functional group, -NCO will produce polyurethanes.
Polyurethane • Polyurethanes are somehow considered as addition polymers since there is no small molecules formed during the reaction. However there is no urethane monomers that add together to form polyurethanes. • Polyurethanes are made in situ and moulded into shape wanted. • (“In the reaction mixture." There are numerous unstable molecules which must be synthesized in situ (i.e. in the reaction mixture but cannot be isolated on their own )
Properties of polymers • Properties of condensation polymers depend on their structures. • The benzene ring bonds in more than one position of phenol-methanal plastics forms 3-D cross-linked structure
Properties • The structure gives high strength. Therefore the compound is • High melting point ( decompose before melting) • Unreactive • High electrical resistance • It can be use as an ingredient in worktops and printed-circuit-board insulation • Before it was use as the casing for radios
Kevlar • Kevlar is a polyamide • It forms when 1,2-diaminobenzene (paraphenylenediamine) condenses with benzene-1,4-dicarbonyl chloride(terephthaloyl dichloride)
Long chain • Further polymerization forms a strong three-dimensional structure because of the hydrogen bonding between the long rigid chain • Kevlar is used to produce lightweight bullet-proof vest, composites for motor-cycle helmets and amour.
Modification of polymers • Apart from adding plasticizers (covered in addition polymer) there are 3 more ways to modify polymers • Blowing air into polyurethane to make polyurethane foams ( cushions and thermal insulator) • Blending fibres of polyesters (PET) with other manufactured or natural fibres to make clothes become dye-fast and comfortable than pure polyesters.
Modifying polymer • Another way is adding chemicals ( this process is called doping polymer) such as • Iodine which removes electrons or • Alkali metals which adds electrons • When ethyen is polymerized, the cis-form of polyethene is a electrical conductor due to the deloclization of the pi electrons. • The conductivity is increaseddramatically after doping I2
Advantages • Use to produce lightweight bullet-proof vest, composites for motor-cycle helmets and amour. (PET) • Devices such as billiard balls, dominoes, Mahjongg tiles and other gaming tilesets, and movers/pieces for games like chess, checkers, and backgammon, are constructed of Bakelite(phenol-methanal plastics) for the look, durability, fine polish, weight, and sound of the resulting pieces • Polyurethane products have many uses. Over three quarters of the global consumption of polyurethane products is in the form of foams, with flexible and rigid types being roughly equal in market size. In both cases, the foam is usually behind other materials: flexible foams are behind upholstery fabrics in commercial and domestic furniture; rigid foams are inside the metal and plastic walls of most refrigerators and freezers, or behind paper, metals and other surface materials in the case of thermal insulation panels in the construction sector.
Disadvantages • The disadvantages of condensation polymer are similar to addition polymer • Low-reactivity Not easy to dispose off. • Generally not biodegradable • Produce hydrogen cyanide gas when polyurethanes are burned.