Catalyst
E N D
Presentation Transcript
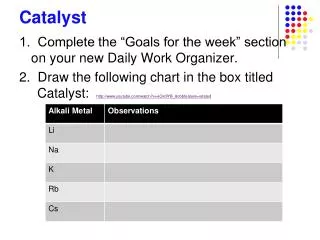
Catalyst • Label the following as physical or chemical changes: • Burning a candle • Cracking an egg • Melting Butter • Freezing ice • Aluminum foil reacting with pool cleaner
Unit 1 – Atomic Theory, The Periodic Table, and Food Toxicology Dangerous Elements
It takes 31 cuts to get to the size of the atom! It is 0.0000007 mm!
Atom’s Family • a
Today’s Learning Target LT 1.1 – I can identify the size, location, and mass of the three subatomic particles found in the atom.
At the ParticulateLevel(APL) • Now that we’ve talked about particles as atoms, molecules, elements, compounds, and mixtures, let’s focus on each on individually. • What is in our food at the smallest level?
Size of the Atom • The atom has a diameter between 0.1 – 0.5 nm • Point of reference: A basketball has a diameter of 234,500,000 nm
Three Types of Subatomic Particles • All atoms contain the following subatomic particles: • Protons • Neutrons • Electrons
Protons Protonsare found in the nucleus of an atom. The nucleus is located in the center of the atom. Protons have a positive charge. We say that their charge is +1
Neutrons James Chadwick discovered the neutron in 1932! Neutronsare found in the nucleus of an atom. Neutrons do not have a charge. They are neutral
Mass of Protons and Neutrons • Protons and neutrons have the same mass. • Each subatomic particle has a mass of 1 amu. • amu= atomic mass unit
Electrons Electronsare found in the electron cloud. The electron cloud surrounds the nucleus of an atom. Electrons have a -1 charge
Mass of Electrons Electrons have a mass that is 0.00054 amu. Electrons do not contribute to the weight of an atom.
Neutral • Like the car gear, like water, like switzerland! To be neutral, the positives and negatives must cancel out! Neutral Tweet! Equal nmbr of protons and electrons #Chemswag #Learning #MrAstorIsABoss The resulting charge is zero!
Scientific Literacy - Death by Element • Complete the reading using your Curriculet log-ins • You must answer the appropriate questions to advance in the reading.
Rate Yourself • Based on the exit ticket and your current level of understanding, rate yourself 1 – 4 on LT 1.1
Closing Time • No practice!