Review of Human Mitochondrial Proteins MTHFD1L and MTHFD2L: Functions and Medical Relevance
This document reviews human mitochondrial proteins MTHFD1L and MTHFD2L, focusing on their functions and medical significance. MTHFD1L is involved in ATP binding, forms a homodimer, and interacts with eleven other proteins. It has crucial substrates such as ATP and tetrahydrofolate, producing ADP and 10-formyltetrahydrofolate. MTHFD2L's features and substrate interactions are also discussed. The suggestions for future research include obtaining boundaries for soluble proteins, guided by Dean Appling, and awaiting results from NESG screenings.


Review of Human Mitochondrial Proteins MTHFD1L and MTHFD2L: Functions and Medical Relevance
E N D
Presentation Transcript
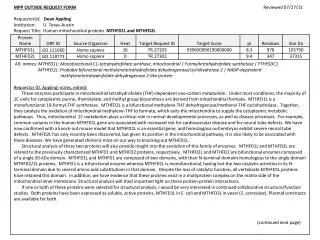
MPP OUTSIDE REQUEST FORM Reviewed 07/27/11 Requestor(s): Dean Appling Institution: U. Texas-Austin Request Title: Human mitochondrial proteins MTHFD1L and MTHFD2L
MPP notes (dja): MTHFD1L Function and Medical Relevance - MTHFD1L is an MPP "ListB" (Most) Biological Relevance target. See reviewer notes. Physical Characteristics-The 978 aa sequence features a transit peptide (1-31) that is cleaved to create the mature protein, an ATP-binding region (423-430), an inactive MTHFD /cyclohydrolase domain (31-348), and an FTHF synthetase domain (349-978). This is the canonical isoform; isoform 2 is much smaller (30 kda). Two lysines are acetylated. MTHFD1L is said to form a homodimer. Similar Structures-A bacterial N(10)-formyltetrahydrofolate synthetase aligning with 53% identity over aa 362-976 has been solved (1eg7a). Interactors-11 other proteins have been demonstrated to bind to MTHFD1L. Substrates are ATP, formate, tetrahydrofolate; products are ADP, phosphate , and 10-formyltetrahydrofolate. Kms for THF monoglutamate, triglutamate, and pentaglutamate have been determined. Status- MTHFD1L was selected in List B by MPP but not yet placed in a cloning workgroup. In June 2011, NESG cloned multiple variants encompassing 4 domains or fragments, approximately residues 65-350, 72-181, 182-298, and 372-968. MTHFD2L Function and Medical Relevance - See reviewer notes. Physical Characteristics-The 347 aa sequence sent by Dean Appling is 58 aa longer at the N-term than the longest isoform in Uniprot , otherwise they match. TargetP confidently predicts a transit peptide (1-8), Uniprot does not predict one. Functional domains are predicted by PFAM (1-111 and 114-288). There are two smaller alternative splicing isoforms. Similar Structures-There are 3 prokaryotic structures with 47-49% identity over most of the length of the protein, and a human folate-dependent dehydrogenase/cyclohydrolase bifunctional enzyme with 42% identity (1a4iA) with NADPH bound. Interactors-Substrates include NAD, products NADH; Mg2+ is likely to be a cofactor. Status - MPP has not yet selected this protein. NESG selected it in May 2011 and designed constructs of residues 1-112 and 113-289. Suggestions 1. Get boundaries from Dean Appling for soluble, active proteins he mentions. Pass info to NESG, and wait for results from their screens before doing any MPP work. Conclusions – suggestions accepted. Dja will contact Dean Appling, get boundaries, compare to NESG fragments, check on expression before MPP does any work.