Properties and classification of matter
1.3k likes | 1.98k Vues
Properties and classification of matter. What is matter?. All matter is composed of particles ( atoms , ions or molecules ). 6. We will be studying states of matter: solids liquids gases There is also plasma, liquid crystal and Bose-Einstein condensates. Mass.

Properties and classification of matter
E N D
Presentation Transcript
All matter is composed of particles (atoms, ions or molecules).
6 • We will be studying states of matter: • solids • liquids • gases • There is also plasma, liquid crystal and Bose-Einstein condensates
Mass – the quantity of particles in a substance - mesured in kg or pounds –the quantity of space occupied by a substance - mesured in cm3 or mL Volume Solids Liquids
SOLIDS • The particles are very close together • There is a high force of attraction between the particles
SOLIDS • Possess a definedmass (does not change) Possess a definedform (does not change)
SOLIDS • The particles don’t spread out in a container • Cannot be invisible The particles don’t move around, but they can vibrate. The force of gravity is always pushing on the particles.
LIQUIDS • The particles are close together, but there is space between them. The force of attraction between the particles is less than that of solids. The particles can easily move around.
LIQUIDS • Possess a defined mass Possess a defined volume
LIQUIDS • The particles spread out in a container (they take the container’s form, so their form is indefinite). • Cannot be invisible Particles can move easily
GASES • The particles are very spread out. • Possess an undefined volume. The force of attraction is very small, as is the force of gravity.
GASES • Possess an undefinedmass (as the particles are always moving) Possess an undefinedform (take the form of their container) ???
GASES • The particles spread out in a container (take its form) • Can be invisible The particles can easily move around
Review: Solids • Mass? • Volume? • Form? • Attraction? • Particles? • Invisible? • Take the form of the container? defined defined defined high close together no no
Review: Liquids defined • Mass? • Volume? • Form? • Attraction? • Particles? • Invisible? • Take the form of the container? defined undefined somewhat attracted somewhat spaced out no yes
Review:Gases • Mass? • Volume? • Form? • Attraction? • Particles? • Invisible? • Take the form of the container? undefined undefined undefined a little spread out yes/no yes
Review: Solid, Liquid and/or Gas??
Review: Solid, Liquid and/or Gas?? Particles move quickly Gas
Review: Solid, Liquid and/or Gas?? Particles only vibrate Solid
Review: Solid, Liquid and/or Gas?? Takes the form of its container Gas and Liquid
Review: Solid, Liquid and/or Gas?? The particles are very spread out Gas
Review: Solid, Liquid and/or Gas?? Defined mass Solid and Liquid
Review: Solid, Liquid and/or Gas?? Very little attraction between the particles Gas
Review: Solid, Liquid and/or Gas?? Does not take the form of its container Solid
Review: Solid, Liquid and/or Gas?? Defined masse and volume Solid and Liquid
Review: Solid, Liquid and/or Gas?? Not affected by gravity Gas
Review: Solid, Liquid and/or Gas?? Defined masse, defined volume, undefined form Liquid
Review: Solid, Liquid and/or Gas?? Undefined Volume Gas
Review: Solid, Liquid and/or Gas?? Strong attraction between particles Solid
Review: Solid, Liquid and/or Gas?? Particles can move around Gas and Liquid
Review: Solid, Liquid and/or Gas?? Defined masse, defined volume, defined form Solid
The particle theory of matter - See page 156 of Science Power 9
Plasma • Exists at very high temperatures • Exists in nature • Used in atomic lasers • Examples: • The Sun’s nucleus • Stars • Flashes of lightning
Bose-Einstein Condensates • The lowest state of matter • Exists at very cold termperatures only • Absolute Zero (0°Kelvin = -273 °Celsius) • All particle movement stops • A SUPER SOLID is formed!!
Bose-Einstein Condensate
CONDENSATION SOLIDIFICATION SUBLIMATION EVAPORATION MELTING Bose-Einstein Condensate
Properties of Matter
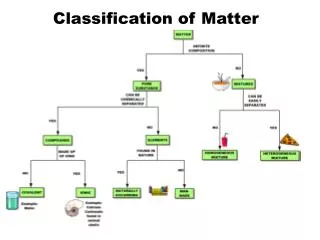
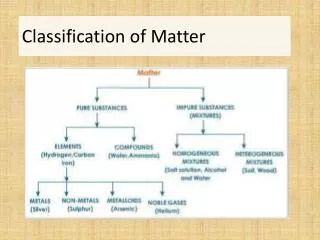
Matter Mixtures Pure Substances
Pure Substances • A pure substance pure is made up of one type of particle (atoms or molecules) • Water • Chlorine • Oxygen
Mixtures • Contain 2+ different types of particles that do not chemically react. They maintain their chemical characteristics but their physical properties may change. • Can be heterogenous or homogenous • Examples: • Sugar and water • Salt and pepper • Gold ring • Diet Coke
Pure substances Mixture Element Compound Homogeneous Heterogeneous All particles are identical. There is more than one type of particle
Matter Mixtures Pure Substances Mechanical Mixtures Solutions
Classify the Mixture
Classify the Mixture DietPepsi Heterogenous