Radioactive Decay
E N D
Presentation Transcript
Radioactivity vs. chemical reactions • CHEMICAL REACTIONS make new substances by rearranging atoms and forming bonds. The atoms do not change in the process. • NUCLEAR REACTIONS instead involve a change in the nucleus and changes the atom…forming a different element.
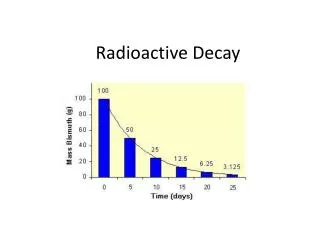
What is Radioactivity? • By late 1890s scientists noticed some substances spontaneously emitted radiation and called it RADIOACTIVITY. • The particles emitted were called RADIATIONwhere atoms change their identity in the process. • Radioactivity happens because of unstable nuclei break down to more stable forms. • This process is called RADIOACTIVE DECAY.
TYPES OF RADIATION? 1) ALPHA: Each alpha particle emits 2 protons and 2 neutrons. What element is this equivalent to??? If Radium (Ra) -226 which has an atomic number of 88 and undergoes a decay of an alpha particle, how many protons will the new element have? What element will it turn into? What will the resulting mass be?
BETA RADIATION: • Radiation that consists of fast moving negative particles called BETA PARTICLES. • MASS OF THE ELEMENT DOES NOT CHANGE. • THE ATOMIC NUMBER INCREASES BY 1. • IF Carbon 14 has an atomic number of 6, and a beta particle was released… - WHAT WOULD THE RESULTING ELEMENT BE?
Gamma Radiation • High energy radiation that has no mass and no electric charge
COOL NUCLEAR SITES • http://people.chem.duke.edu/~jds/cruise_chem/nuclear/nuclear.htm • Where would you like to go to learn more??? • Radioactivity : The Pros and Cons • Everyday Exposure to Radiation • Modern Uses of Radioactive Isotopes • Detection of Radiation and your Protection • The Discovery of Radioactivity • Nuclear Stability and Radioactive Decay • Other Radioisotopes Found in Everyday Life • Shedding Light on Some Common Misconceptions and Fears • Social, Economic, and Political Concerns