Chapter 6
Chapter 6. How cells harvest chemical energy. Musebio101 summer 2010. Photosynthesis and cellular respiration provide energy for life. Energy is necessary for life processes These include growth, transport, manufacture, movement, reproduction, and others

Chapter 6
E N D
Presentation Transcript
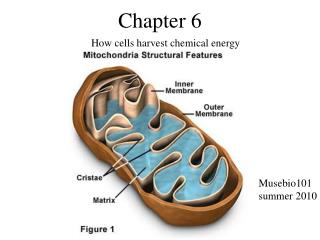
Chapter 6 How cells harvest chemical energy Musebio101 summer 2010
Photosynthesis and cellular respiration provide energy for life • Energy is necessary for life processes • These include growth, transport, manufacture, movement, reproduction, and others • Energy that supports life on Earth is captured from sun rays reaching Earth through plant, algae, protist, and bacterial photosynthesis
Sunlight energy ECOSYSTEM Photosynthesis in chloroplasts Glucose CO2 + + O2 H2O Cellular respiration in mitochondria ATP (for cellular work) Heat energy
Cellular respiration banks energy in ATP molecules • Cellular respiration is an exergonic process that transfers energy from the bonds in glucose to ATP • Cellular respiration produces 38 ATP molecules from each glucose molecule • Other foods (organic molecules) can be used as a source of energy as well
Introduction to Metabolism • Cells break down organic molecules to obtain energy • Used to generate ATP • Most energy production takes place in mitochondria
Metabolism • Metabolism – refers to all chemical reaction occurring in body • Catabolism – break down complex molecules • Exergonic – produce more energy than they consume • Anabolism – combine simple molecules into complex ones • Endergonic – consume more energy than they produce • Adenosine triphosphate (ATP) • “energy currency” • ADP + P + energy ↔ ATP
Reactants Amount of energy released Potential energy of molecules Energy released Products
Metabolism • Catabolism • Is the breakdown of organic substrates • Releases energy used to synthesize high-energy compounds (e.g., ATP) • Anabolism • Is the synthesis of new organic molecules
Cellular respiration is a catabolic reaction + CO2 H2O C6H12O6 6 ATPs + 6 O2 + 6 Carbon dioxide Glucose Oxygen Water Energy
Waste products Energy conversion Fuel Heat Cellular respiration Carbon dioxide Glucose Oxygen Water Energy for cellular work Energy conversion in a cell
Phosphorylation Hydrolysis Energy for endergonic reactions Energy from exergonic reactions
Adenosine Triphosphate (ATP) Phosphate group Adenine Ribose Hydrolysis + Diphosphate (ADP) Adenosine
Carbohydrate Metabolism • Mitochondrial Membranes • Outer membrane • Contains large-diameter pores • Permeable to ions and small organic molecules (pyruvic acid) • Inner membrane • Contains carrier protein • Moves pyruvic acid into mitochondrial matrix • Intermembrane space • Separates outer and inner membranes
Chemical energy (high-energy electrons) Chemical energy Electron transport chain and oxidative phosphorylation Glycolysis Krebs cycle Pyruvic acid Glucose Mitochondrial cristae Mitochondrion Cytosol Via oxidative phosphorylation Via substrate-level phosphorylation 1 2 3 During glycolysis, each glucose molecule is broken down into two molecules of pyruvic acid in the cytosol. Energy-rich electrons picked up bycoenzymes are transferred to the electron transport chain, built into the cristae membrane. The electron transport chain carries out oxidative phosphorylation, which accounts for most of the ATP generated by cellular respiration. The pyruvic acid then enters the mitochondrial matrix, where the Krebs cycle decomposes it to CO2. During glycolysis and the Krebs cycle, small amounts of ATP are formed by substrate- level phosphorylation. Figure 24.5
Carbohydrate Metabolism • Glucose Breakdown • Occurs in small steps • Which release energy to convert ADP to ATP • One molecule of glucose nets 36 molecules of ATP • Glycolysis • Breaks down glucose in cytosol into smaller molecules used by mitochondria • Does not require oxygen: anaerobic reaction • Aerobic Reactions • Also called aerobic metabolism or cellular respiration • Occur in mitochondria, consume oxygen, and produce ATP
Catalysis Enzyme Enzyme (a) Substrate-level phosphorylation
ENERGY INVESTMENT PHASE Glucose ATP Steps – A fuel molecule is energized, using ATP. Step 1 3 1 ADP Glucose-6-phosphate P 2 P Fructose-6-phosphate ATP 3 ADP P P Fructose-1,6-bisphosphate Step A six-carbon intermediate splits Into two three-carbon intermediates. 4 4 Glyceraldehyde-3-phosphate (G3P) P P NAD+ NAD+ Step A redox reaction generates NADH. 5 5 ENERGY PAYOFF PHASE 5 P P NADH NADH + H+ + H+ P P P P 1,3-Bisphosphoglycerate ADP ADP 6 6 ATP ATP P P 3-Phosphoglycerate 7 7 Steps – ATP and pyruvate are produced. P P 6 9 2-Phosphoglycerate 8 8 H2O H2O P P Phosphoenolpyruvate (PEP) ADP ADP 9 9 ATP ATP Pyruvate
H C H C H C H C H C H C O O O O O O P P P P P P CH2O CH2O CH2O CH2O CH2O CH2O C C C C C C O O O O O O HCOH HCOH HCOH HCOH HCOH HCOH 6 6 6 6 6 6 6 6 6 6 CH2OH CH2OH CH2OH CH2OH CH2OH CH2OH CH2OH CH2OH CH2OH CH2OH CH2OH CH2OH CH2OH CH2OH CH2OH CH2OH CH2O CH2O CH2O CH2O CH2O CH2O P P P P P P 5 5 5 5 5 5 5 5 5 5 O O O O O O O O O O H H H H H H H H H H H H H H H H H H H H Dihydroxyacetone phosphate Dihydroxyacetone phosphate Dihydroxyacetone phosphate Dihydroxyacetone phosphate Dihydroxyacetone phosphate Dihydroxyacetone phosphate Glyceraldehyde 3-phosphate Glyceraldehyde 3-phosphate Glyceraldehyde 3-phosphate Glyceraldehyde 3-phosphate Glyceraldehyde 3-phosphate Glyceraldehyde 3-phosphate H H H H H H H H H H 5 5 5 5 5 5 4 4 4 4 4 4 4 4 4 4 1 1 1 1 1 1 1 1 1 1 H H H H H H H H H H OH OH OH OH OH OH OH OH OH OH HO HO HO HO HO HO HO HO HO HO OH OH OH OH OH OH OH OH OH OH 2 NAD+ 2 NAD+ 2 NAD+ 2 NAD+ 2 NAD+ + 2 + 2 + 2 + 2 + 2 P P P P P 6 6 6 6 6 3 3 3 3 3 3 3 3 3 3 2 2 2 2 2 2 2 2 2 2 OH OH OH OH OH OH OH OH OH OH Glucose (1 molecule) Glucose (1 molecule) Glucose (1 molecule) Glucose (1 molecule) Glucose (1 molecule) Glucose (1 molecule) Glucose (1 molecule) Glucose (1 molecule) Glucose (1 molecule) Glucose (1 molecule) H H H H H H H H H H NADH NADH NADH NADH NADH + 2H+ + 2H+ + 2H+ + 2H+ + 2H+ 2 2 2 2 2 CH2O CH2O CH2O CH2O CH2O P P P P P ATP ATP ATP ATP ATP ATP ATP ATP ATP ATP HCOH HCOH HCOH HCOH HCOH 1 1 1 1 1 1 1 1 1 1 ADP ADP ADP ADP ADP ADP ADP ADP ADP ADP 1, 3-Bisphosphoglyceric acid (2 molecules) 1, 3-Bisphosphoglyceric acid (2 molecules) 1, 3-Bisphosphoglyceric acid (2 molecules) 1, 3-Bisphosphoglyceric acid (2 molecules) 1, 3-Bisphosphoglyceric acid (2 molecules) C C C C C O O O O O P P P P P O O O O O OH2C OH2C OH2C OH2C OH2C OH2C OH2C OH2C OH2C P P P P P P P P P 2 ADP 2 ADP 2 ADP 2 ADP O O O O O O O O O 7 7 7 7 H H H H H H H H H H H H H H H H H H ATP ATP ATP ATP H H H H H H H H H 2 2 2 2 OH OH OH OH OH OH OH OH OH H H H H H H H H H P P P P CH2O CH2O CH2O CH2O HO HO HO HO HO HO HO HO HO OH OH OH OH OH OH OH OH OH HCOH HCOH HCOH HCOH 3-Phosphoglyceric acid (2 molecules) 3-Phosphoglyceric acid (2 molecules) 3-Phosphoglyceric acid (2 molecules) 3-Phosphoglyceric acid (2 molecules) H H H H H H H H H Glucose 6-phosphate Glucose 6-phosphate Glucose 6-phosphate Glucose 6-phosphate Glucose 6-phosphate Glucose 6-phosphate Glucose 6-phosphate Glucose 6-phosphate Glucose 6-phosphate OH OH OH OH OH OH OH OH OH COOH COOH COOH COOH 2 2 2 2 2 2 2 2 2 8 8 8 6 6 6 6 6 6 6 6 OH2C OH2C OH2C OH2C OH2C OH2C OH2C OH2C P P P P P P P P O O O O O O O O CH2OH CH2OH CH2OH 1 1 1 1 1 1 1 1 CH2OH CH2OH CH2OH CH2OH CH2OH CH2OH CH2OH CH2OH HCO HCO HCO P P P 2-Phosphoglyceric acid (2 molecules) 2-Phosphoglyceric acid (2 molecules) 2-Phosphoglyceric acid (2 molecules) 5 5 5 5 5 5 5 5 2 2 2 2 2 2 2 2 H H H H H H H H HO HO HO HO HO HO HO HO COOH COOH COOH H H H H H H H H OH OH OH OH OH OH OH OH 4 4 4 4 4 4 4 4 3 3 3 3 3 3 3 3 9 9 OH OH OH OH OH OH OH OH H H H H H H H H Fructose 6-phosphate Fructose 6-phosphate Fructose 6-phosphate Fructose 6-phosphate Fructose 6-phosphate Fructose 6-phosphate Fructose 6-phosphate Fructose 6-phosphate CH2 CH2 ATP ATP ATP ATP ATP ATP ATP ATP Phosphoenolpyruvic acid (2 molecules) Phosphoenolpyruvic acid (2 molecules) C C O O P P Phosphofructokinase Phosphofructokinase Phosphofructokinase Phosphofructokinase Phosphofructokinase Phosphofructokinase Phosphofructokinase Phosphofructokinase 3 3 3 3 3 3 3 3 ADP ADP ADP ADP ADP ADP ADP ADP COOH COOH 2 ADP P P P P P P P OH2C OH2C OH2C OH2C OH2C OH2C OH2C O O O O O O O 10 CH2O CH2O CH2O CH2O CH2O CH2O CH2O P P P P P P P 2 ATP H H H H H H H HO HO HO HO HO HO HO H H H H H H H OH OH OH OH OH OH OH CH 3 C O OH OH OH OH OH OH OH H H H H H H H Fructose 1, 6-bisphosphate Fructose 1, 6-bisphosphate Fructose 1, 6-bisphosphate Fructose 1, 6-bisphosphate Fructose 1, 6-bisphosphate Fructose 1, 6-bisphosphate Fructose 1, 6-bisphosphate Pyruvic acid (2 molecules) COOH 4 4 4 4 4 4 4
Glucose 2 NAD+ 2 ADP P 2 GLYCOLYSIS 2 ATP NADH 2 2 Pyruvate NADH 2 CO2 2 released 2 NAD+ 2 Ethanol Alcohol fermentation
Glycolysis evolved early in the history of life on Earth • Glycolysis is the universal energy-harvesting process of living organisms • So, all cells can use glycolysis for the energy necessary for viability • The fact that glycolysis has such a widespread distribution is good evidence for evolution
Carbohydrate Metabolism • Oxidation and Reduction • Oxidation (loss of electrons) • Electron donor is oxidized • Reduction (gain of electrons) • Electron recipient is reduced • The two reactions are always paired
Energy transfer • Oxidation-reduction or redox reactions • Oxidation – removal of electrons • Decrease in potential energy • Dehydrogenation – removal of hydrogens • Liberated hydrogen transferred by coenzymes • Nicotinamide adenine dinucleotide (NAD) • Flavin adenine dinucleotide (FAD) • Glucose is oxidized • Reduction – addition of electrons • Increase in potential energy
Carbohydrate Metabolism • The TCA Cycle (citric acid cycle) • The function of the citric acid cycle is • To remove hydrogen atoms from organic molecules and transfer them to coenzymes • In the mitochondrion • Pyruvic acid reacts with NAD and coenzyme A (CoA) • Producing 1 CO2, 1 NADH, 1 acetyl-CoA • Acetyl group transfers • From acetyl-CoA to oxaloacetic acid • Produces citric acid
Acetyl CoA CoA CoA 2 CO2 CITRIC ACID CYCLE 3 NAD+ FADH2 3 NADH FAD 3 H+ P ADP + ATP
Cytosol Electron trans- port chain and oxidative phosphorylation Glycolysis Krebs cycle Pyruvic acid from glycolysis NAD+ CO2 Transitional phase NADH+H+ Acetyl CoA Carbon atom Mitochondrion (matrix) Inorganic phosphate Oxaloacetic acid Coenzyme A Citric acid (pickup molecule) (initial reactant) NADH+H+ NAD+ Isocitric acid Malic acid NAD+ Krebs cycle CO2 NADH+H+ -Ketoglutaric acid Fumaric acid CO2 NAD+ FADH2 Succinic acid Succinyl-CoA NADH+H+ FAD GDP + GTP ADP
Carbohydrate Metabolism • The TCA Cycle • CoA is released to bind another acetyl group • One TCA cycle removes two carbon atoms • Regenerating 4-carbon chain • Several steps involve more than one reaction or enzyme • H2O molecules are tied up in two steps • CO2 is a waste product • The product of one TCA cycle is • One molecule of GTP (guanosine triphosphate)
CO2 CO2 CO2 CO2 CO2 CO2 CO2 CO2 CoA CoA CoA CoA CoA CoA CoA CoA CH3 CH3 CH3 CH3 CH3 CH3 CH3 CH3 C C C C C C C C O O O O O O O O C C C C C C C C O O O O O O O O CH3 CH3 CH3 CH3 CH3 CH3 CH3 CH3 COOH COOH COOH COOH COOH COOH COOH COOH NADH NADH NADH NADH NADH NADH NADH NADH + H+ + H+ + H+ + H+ + H+ + H+ + H+ + H+ NAD+ NAD+ NAD+ NAD+ NAD+ NAD+ NAD+ NAD+ Pyruvicacid Pyruvicacid Pyruvicacid Pyruvicacid Pyruvicacid Pyruvicacid Pyruvicacid Pyruvicacid Acetyl coenzyme A Acetyl coenzyme A Acetyl coenzyme A Acetyl coenzyme A Acetyl coenzyme A Acetyl coenzyme A Acetyl coenzyme A Acetyl coenzyme A To electron transport chain To electron transport chain To electron transport chain To electron transport chain To electron transport chain To electron transport chain To electron transport chain To electron transport chain Oxaloacetic acid Oxaloacetic acid Oxaloacetic acid Oxaloacetic acid Oxaloacetic acid Oxaloacetic acid Oxaloacetic acid Oxaloacetic acid CoA CoA CoA CoA CoA CoA CoA CoA COOH COOH COOH COOH COOH COOH COOH COOH C C C C C C C C O O O O O O O O NADH + H+ H2C H2C H2C H2C H2C H2C H2C H2C COOH COOH COOH COOH COOH COOH COOH COOH CH2 CH2 CH2 CH2 CH2 CH2 CH2 CH2 HOC HOC HOC HOC HOC HOC HOC HOC COOH COOH COOH COOH COOH COOH COOH COOH 1 1 1 1 1 1 1 1 COOH COOH COOH COOH COOH COOH COOH COOH NAD+ H2O H2O H2O H2O H2O H2O H2O H2O Citric acid Citric acid Citric acid Citric acid Citric acid Citric acid Citric acid Citric acid H2C H2C H2C H2C H2C H2C H2C H2C COOH COOH COOH COOH COOH COOH COOH COOH COOH COOH 8 HCOH HCOH CH2 CH2 2 2 2 2 2 2 2 To electron transport chain To electron transport chain To electron transport chain COOH COOH Malic acid Malic acid H2O H2O 7 7 H2C H2C H2C H2C H2C H2C H2C COOH COOH COOH COOH COOH COOH COOH HC HC HC HC HC HC HC COOH COOH COOH COOH COOH COOH COOH COOH COOH COOH KREBS CYCLE KREBS CYCLE KREBS CYCLE KREBS CYCLE KREBS CYCLE KREBS CYCLE KREBS CYCLE KREBS CYCLE CH CH CH HOC HOC HOC HOC HOC HOC HOC COOH COOH COOH COOH COOH COOH COOH Fumaric acid Fumaric acid Fumaric acid HC HC HC Isocitric acid Isocitric acid Isocitric acid Isocitric acid Isocitric acid Isocitric acid Isocitric acid H H H H H H H COOH COOH COOH 3 3 3 3 3 3 FADH2 FADH2 FADH2 6 6 6 FAD FAD FAD NAD+ NAD+ NAD+ NAD+ NAD+ NAD+ H2C H2C H2C H2C COOH COOH COOH COOH CO2 CO2 CO2 CO2 CO2 CO2 CoA CoA CoA CoA H2C H2C H2C H2C COOH COOH COOH COOH NADH NADH NADH NADH NADH NADH + H+ + H+ + H+ + H+ + H+ + H+ Succinic acid Succinic acid Succinic acid Succinic acid 5 5 5 5 H2C H2C H2C H2C H2C H2C COOH COOH COOH COOH COOH COOH CO2 CO2 CO2 CO2 CO2 GTP GTP GTP GTP 4 4 4 4 4 HCH HCH HCH HCH HCH HCH H2C H2C H2C H2C H2C COOH COOH COOH COOH COOH GDP GDP GDP GDP O O O O O O C C C C C C COOH COOH COOH COOH COOH COOH ADP ADP ADP ADP CH2 CH2 CH2 CH2 CH2 CoA CoA CoA CoA CoA O O O O O C C C C C S S S S S Alpha-ketoglutaric acid Alpha-ketoglutaric acid Alpha-ketoglutaric acid Alpha-ketoglutaric acid Alpha-ketoglutaric acid Alpha-ketoglutaric acid ATP ATP ATP ATP Succinyl CoA Succinyl CoA Succinyl CoA Succinyl CoA Succinyl CoA NAD+ NAD+ NAD+ NAD+ NAD+ To electron transport chain To electron transport chain To electron transport chain To electron transport chain To electron transport chain To electron transport chain NADH NADH NADH NADH NADH + H+ + H+ + H+ + H+ + H+
Carbohydrate Metabolism A Summary of the Energy Yield of Aerobic Metabolism.
Carbohydrate Metabolism • Summary: The TCA Cycle (Krebs cycle) CH3CO - CoA + 3NAD + FAD + GDP + Pi + 2 H2O CoA + 2 CO2 + 3NADH + FADH2 + 2 H+ + GTP
Carbohydrate Metabolism • The Electron Transport System (ETS) • Is the key reaction in oxidative phosphorylation • Is in inner mitochondrial membrane • Electrons carry chemical energy • Within a series of integral and peripheral proteins
Carbohydrate Metabolism • Coenzyme FAD • Accepts two hydrogen atoms from TCA cycle: • Gaining two electrons • Coenzyme NAD • Accepts two hydrogen atoms • Gains two electrons • Releases one proton • Forms NADH + H+
1 Glucose 1 Glucose 1 Glucose 1 Glucose 2 2 2 2 ATP ATP ATP ATP 1 1 1 1 GLYCOLYSIS GLYCOLYSIS GLYCOLYSIS GLYCOLYSIS 2 2 2 2 NADH NADH NADH NADH + 2 H+ + 2 H+ + 2 H+ + 2 H+ 2 Pyruvic acid 2 Pyruvic acid 2 Pyruvic acid 2 Pyruvic acid 2 2 2 FORMATION OF ACETYL COENZYME A FORMATION OF ACETYL COENZYME A FORMATION OF ACETYL COENZYME A CO2 CO2 CO2 2 2 2 2 2 2 NADH NADH NADH + 2 H+ + 2 H+ + 2 H+ 4 ELECTRON TRANSPORT CHAIN 2 Acetyl coenzyme A 2 Acetyl coenzyme A 2 Acetyl coenzyme A Electrons 32 or 34 ATP 2 2 ATP ATP e– 4 4 CO2 CO2 e– 3 3 e– 6 6 NADH NADH KREBS CYCLE KREBS CYCLE + 6 H+ + 6 H+ 6 O2 2 2 FADH2 FADH2 H2O 6
NADH ATP NAD+ + 2e– Controlled release of energy for synthesis of ATP H+ Electron transport chain 2e– O2 1 2 H+ H2O
Carbohydrate Metabolism Oxidative Phosphorylation.
NADH+H+ Electron trans- port chain and oxidative phosphorylation Glycolysis Krebs cycle FADH2 Enzyme Complex II Enzyme Complex I Enzyme Complex III Free energy relative to O2 (kcal/mol) Enzyme Complex IV
Space between outer and inner mitochondrial membranes H+channel H+ + + + + + + + H+ H+ Cyt c Inner mito- chondrial membrane e– e– Q e– e– e– – – – – – – – Mitochondrial matrix 3 O2 1 1/2 NAD + H+ NADH H+ ADP + P 3 H2O ATP ATP synthase 1 2 3 NADH dehydrogenase complex: FMN and five Fe-S centers Cytochrome b-c1 complex: cyt b, cyt c1, and an Fe-S center Cytochrome oxidase complex: cyt a, cyt a3,and two Cu The actions of the three proton pumps and ATP synthase in the inner membrane of mitochondria
High H+ concentration in intermembrane space Membrane Proton pumps (electron transport chain) ATP synthase Energy from food ADP + Low H+ concentration in mitochondrial matrix (b) Oxidative phosphorylation
Overview of metabolic processes Stage 1 Digestion in GI tract lumen to absorbable forms. Transport via blood to tissue cells. PROTEINS CARBOHYDRATES FATS Glucose and other sugars Fatty acids Amino acids Glycerol Stage 2 Anabolism (incorporation into molecules) and catabolism of nutrients to form intermediates within tissue cells. Glycogen Glucose Proteins Fats NH3 Pyruvic acid Acetyl CoA Stage 3 Oxidative breakdown of products of stage 2 in mitochondria of tissue cells. CO2 is liberated, and H atoms removed are ultimately delivered to molecular oxygen, forming water. Some energy released is used to form ATP. Krebs cycle Infrequent CO2 O2 Oxidative phosphorylation (in electron transport chain) H2O H
Chemical energy (high-energy electrons) Chemical energy Electron transport chain and oxidative phosphorylation Glycolysis Krebs cycle Pyruvic acid Glucose Mitochondrial cristae Mitochondrion Cytosol Via oxidative phosphorylation Via substrate-level phosphorylation Energy-rich electrons picked up by coenzymes are transferred to the elec- tron transport chain, built into the cristae membrane. The electron transport chain carries out oxidative phosphorylation, which accounts for most of the ATP generated by cellular respiration. 1 2 3 During glycolysis, each glucose molecule is broken down into two molecules of pyruvic acid in the cytosol. The pyruvic acid then enters the mitochondrial matrix, where the Krebs cycle decomposes it to CO2. During glycolysis and the Krebs cycle, small amounts of ATP are formed by substrate- level phosphorylation.
Food, such as peanuts Carbohydrates Fats Proteins Amino acids Glycerol Sugars Fatty acids Amino groups OXIDATIVE PHOSPHORYLATION (Electron Transport and Chemiosmosis) CITRIC ACID CYCLE Acetyl CoA Glucose G3P Pyruvate GLYCOLYSIS ATP
Review • Energy is harvested from high energy glucose by the mitochondria • The process begins in the cytoplasm with glycolysis where glucose is converted to pyruvate • The TCA cycle (Krebs cycle) harvests energy from the pyruvate and stores it as reduced electron carrier molecules • The carrier molecules cash in these electrons for ATP in the electron transport chain if oxygen is available
Review (cont) • 2 net ATP are made from each glucose in glycolysis • 34 additional ATP are made from each glucose if oxygen is available to help run the TCA cycle and electron transport chain