Recombinant DNA Technology
Harini Chandra. Recombinant DNA Technology.

Recombinant DNA Technology
E N D
Presentation Transcript
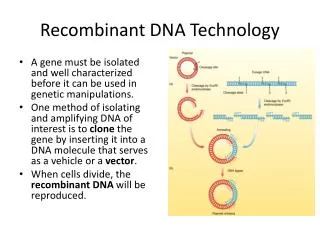
Harini Chandra Recombinant DNA Technology Recombinant DNA technology which came into existence in the 1970s, allows for genetic manipulation of organisms by incorporating DNA sequences from different sources into a single recombinant molecule. This revolutionary technology has opened up several applications in plant genomics and clinical research.
Master Layout (Part 1) 1 This animation consists of 3 parts: Part 1 – Tools used for gene exploration Part 2 – Creation of a genomic library Part 3 – Transgenic plants & animals Polymerase Chain Reaction (PCR) Target sequence Blotting techniques First cycle 2 GAATTC CTTAAG GAATTC CTTAAG Primers Restriction enzyme Taq polymerase Restriction enzyme analysis G G G G AATTC AATTC AATTC AATTC 3 Newly synthesized DNA G G G G CTTAA CTTAA CTTAA CTTAA Second cycle DNA Ligase Restriction sites 4 Probing & autoradiography Cohesive ends Electrophoresis Blotting 5 Agarose gel Autoradiograph Nitrocellulose sheet Source: Biochemistry by Lubert Stryer, 5th & 6th edition (ebook)
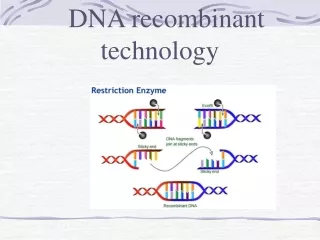
Definitions of the components:Part 1 – Tools used for gene exploration 1 1. Restriction analysis: A molecular biology technique that is used to fragment and manipulate DNA into manageable lengths before further analysis. The following components are required for restriction analysis. a) Restriction enzymes: An enzyme that cleaves single or double stranded DNA at specific locations based on the nucleotide sequences recognized. b) Restriction sites: These are regions of the nucleotide sequence that are recognised by the restriction enzymes and subsequently cleaved. c) DNA Ligase: Enzyme that is involved in repairing or joining single stranded breaks or discontinuities in double stranded DNA. d) Cohesive ends: A cohesive or sticky end of DNA refers to those DNA molecules having a 3’ or 5’ overhang region after they have been cleaved by the restriction enzyme. These overhangs possess nucleotide sequences that have complementary regions and can therefore easily anneal together. e) Blunt ends: Blunt ends of DNA molecules refer to those that do not possess any overhang region but are instead cleaved at the same nucleotide position on both strands thereby producing flat or blunt ends. 2. Blotting techniques: These are techniques that have been developed to characterize DNA and RNA. The Southern blot, developed by Edwin Southern, is used to characterize DNA while the Northern blot is used for RNA. 2 3 4 5
Definitions of the components:Part 1 – Tools used for gene exploration 1 a) Electrophoresis: Electrophoresis is a gel-based analytical technique that is used for separation and visualization of biomolecules like DNA, RNA and proteins based on their fragment lengths or charge-to-mass ratios using an electric field. b) Agarose gel: Gels made of agarose are commonly used for separation of nucleotide molecules. These are made up of 0.7-2% agarose dissolved in the electrophoresis buffer but concentrations can vary depending on the length of fragments to be separated. c) Blotting: The process by which nucleotide molecules separated on the electrophoresis gel are transferred on to another surface such as nitrocellulose by placing them in contact with each other. d) Nitrocellulose sheet: A membrane or sheet made of nitrocellulose onto which single stranded DNA molecules separated by electrophoresis are transferred for further probing and analysis. e) Probing & autoradiography: The single stranded DNA molecules bound to the nitrocellulose membrane are probed for specific sequences by means of a radioactive probe molecule. Excess probe molecules are washed off after which the radioactivity is detected by means of autoradiography. f) Autoradiograph: The image pattern produced due to decay emissions from a radioactive material. 2 3 4 5
Definitions of the components:Part 1 – Tools used for gene exploration 1 3. Polymerase Chain reaction (PCR): The polymerase chain reaction is an extremely useful molecular biology tool that allows for amplification of a segment of DNA upto billion-fold. This helps in characterization and manipulation of very small quantities of DNA and has found several diverse applications. a) Target sequence: The sequence of interest of DNA that is to be amplified. b) PCR cycle: One round or cycle of PCR refers to strand separation at 95oC, annealing of primers at 54oC and elongation of these primers at 72oC by the polymerase enzyme. Every PCR cycle results in doubling of the number of strands present. c) Primers: A short strand of nucleotides that acts as a starting point for DNA synthesis by providing a free 3’-OH end. These are hybridized to the target DNA and then elongated by means of the polymerase. d) Taq polymerase: This is a thermostable DNA Polymerase enzyme that is obtained from the bacterium Thermus aquaticus. It is used for PCR reactions due to its ability to perform DNA elongation even at high temperatures. 2 3 4 5
Part 1, Step 1: 1 Restriction enzyme analysis Cleavage sites 3’ 5’ 3’ 5’ 5’ 5’ G G 3’ G G AATTC AATTC AATTC AATTC 3’ 5’ 2 CTTAA CTTAA CTTAA CTTAA 3’ G G G G EcoR1 Restriction enzyme 3’ 5’ GAATTC CTTAAG GAATTC CTTAAG DNA Ligase DNA sequences 3 3’ 3’ 5’ 5’ 5’ 3’ 3’ 5’ 5’ 3’ 3’ 5’ 4 Action Description of the action Audio Narration First show the green and blue sequences on the left with the red arrows as shown which must be blinking. Then show the pie shaped objects with the label ‘EcoRI’ which must cleave the sequences at the positions indicated to release them as shown in middle panel. Then show the green and blue complementary shapes crossing each other as shown followed by the arrow and the four red circles. These circles must move across the small gaps between the green and blue regions to seal them resulting in the figure as shown on right. As shown in animation. Restriction endonucleases cleave double stranded DNA at specific recognition sequences which can be used for isolating genes and cloning new DNA molecules. These enzymes are typically found in prokaryotes where they function as a defence mechanism against foreign DNA. Recognitions sites are typically 4-8 base pairs long and are very often palindromic i.e. they read the same in both directions. Cleavage at a phosphodiester bond can produce either blunt ends or cohesive ends. Two DNA molecules that have been cleaved by the same restriction enzyme can be ligated using DNA ligase to produce a recombinant molecule. The fragments may also be separated using electrophoresis techniques. 5 Source: Biochemistry by Lubert Stryer, 6th edition (ebook)
Part 1, Step 2: 1 Sample loading Blotting techniques - Cathode Electrophoresis Direction of migration Restriction digest 2 Agarose gel Anode + Buffer Labelled probe molecule 3 Southern blotting P35 Blotting Autoradiography Completed agarose gel Autoradiogram Nitrocellulose sheet 4 Action Description of the action Audio Narration DNA that has been fragmented by restriction enzymes can be separated by agarose gel electrophoresis. The DNA is then denatured and blotted onto a nitrocellulose sheet such that their positions remain intact, a process known as Southern blotting. The DNA strands are then probed with a radiolableled DNA molecule which hybridizes to the complementary sequence and can be detected by autoradiography. A similar procedure for RNA molecules is referred to as Northern blotting. (Please redraw all figures.) First show the figure on top with its labels. The hand must appear and move into one of the wells after which a blue band must appear there & hand must disappear. The arrow on the side with label must appear followed by movement of the blue band in the arrow direction. This blue band must separate into more blue bands with small ones moving down further and larger ones remaining close to top. Next, the grey surface with separated blue bands must be shown below. A paper marked nitrocellulose sheet must be placed on top of this and the blue bands must be blotted on the sheet as well as shown. The red rect must then appear which must bind to one of the blue bands after which the arrow and the label ‘autoradiography’ must appear followed by the figure at right bottom. As shown in animation. 5 Source: Biochemistry by Lubert Stryer, 6th edition (ebook)
Part 1, Step 3 (a): 1 Polymerase Chain Reaction (PCR) Addition of dNTPs & Taq Polymerase Addition of primers First cycle 3’ 5’ Strand separation ~ 95oC(1 min) 2 Annealing ~54oC (45 s) Heating 3’ 5’ Elongation 72oC (2 min) 3 dsDNA sample 4 Action Description of the action Audio Narration The polymerase chain reaction can be used to amplify specific DNA sequences of interest. The reaction consists of multiple cycles of strand separation, primer annealing and strand elongation to generate millions of copies of the target sequence within very short time. The two parent DNA strands are first separated by heating them to 95oC after which large excess of primers are added and the solution cooled abruptly (e.g. 54oC), thereby allowing the primers to anneal to each end of the target strand. Strand elongation then takes place at 72oC with help of heat-stable DNA Polymerase from Thermus aquaticus. Both primers are elongated in 5’ to 3’ direction beyond the target sequence. (Please redraw all figures.) First show the tube on left along with a plate below it. This must be zoomed into and the figure on the right must be shown. The plate must then turn red, indicating heating during which the two strands on the right must separate. Then the hand with pipette must appear & move towards the tube. The green & brown rectangles must appear. Next the hand must again appear like last time. Now the small colored rectangles & two pink ovals must appear. The ovals must move as shown at the end of which, the arrows must appear as depicted. As shown in animation. 5 Source: Biochemistry by Lubert Stryer, 6th edition (ebook)
Part 1, Step 3 (b): 1 Polymerase Chain Reaction (PCR) Addition of dNTPs & Taq Polymerase Addition of primers Second cycle 3’ 5’ Strand separation ~ 95oC(1 min) 2 3’ 5’ Annealing ~54oC (45 s) 3’ 5’ Heating Elongation 72oC (2 min) 3 dsDNA sample 3’ 5’ 4 Action Description of the action Audio Narration Multiple rounds of PCR are carried out in this manner with repeated cycles of strand separation, primer hybridization and strand elongation. It is possible to carry out these sequence of reactions continuously in a closed container without further addition of reagents after the first cycle, simply by modifying the temperature of the reaction mixture. Although the first cycle yields strands that are longer than the target sequence, only the target is amplified from the end of the second cycle onwards. The sequence gets amplified 2n-fold at the end of n cycles. This is an extremely useful technique in diagnostics, forensics and molecular evolution. As shown in animation. (Please redraw all figures.) Same sequence of events as described in previous slide. Sample heating must be shown with the coloured strands separating after which four rectangular boxes must appear when the hand with micropipette appears and moves downwards. The second time, 4 oval circles must be shown which must move across the strands leading to formation of new ones as depicted in animation. 5 Source: Biochemistry by Lubert Stryer, 6th edition (ebook)
Part 1, Step 4 (a): 1 DNA Sequencing – Sanger’s dideoxy method 3’-----GAATTCGCTAATGC------- 5’ Dye-labelled ddNTPs ddATP GCGATT A 5’-----CTTAA 2 ddGTP 3’-----GAATTCGCTAATGC------- 5’ DNA Pol I dCTP, dATP, dGTP, dTTP GCGATTAC G 5’-----CTTAA Template strand 3’-----GAATTCGCTAATGC------- 5’ ddCTP 3’-----GAATTCGCTAATGC------- 5’ 3 5’-----CTTAA Primer GCGATTA C 5’-----CTTAA 3’-----GAATTCGCTAATGC------- 5’ ddTTP GCGA T 5’-----CTTAA 2’-3’ – dideoxy analog 4 Action Description of the action Audio Narration A simple and elegant method for DNA sequencing was devised by Frederick Sanger where a collection of DNA fragments are synthesized by means of controlled interruption of enzymatic replication. Four DNA synthesis reactions are carried out simultaneously with the strand whose sequence is to be determined being used as the template. The reaction mixture consists of regular deoxynucleotides and DNA Polymerase along with a small amount of one labelled dideoxy nucleotide analog being added to each of the four reaction mixtures. A primer is added to begin the DNA synthesis and strand elongation continues until a dideoxy analog gets added instead of the regular dNTP. Chain termination occurs at this stage due to the absence of a 3’ OH group for formation of the next phosphodiester bond. (Please redraw all figures.) First show the blue & red strands on the left with letters followed by the line with text ‘DNA Pol1, dCTP…’. Next the coloured label ‘ddATP’ must be shown on the arrow follwed by the blue & red boxes. The letters in the green box must appear one at a time. Once the ‘A’ in yellow appears, no more letters are added further. Similar sequence is depicted for all four reactions as depicted in animation. As shown in animation. 5 Source: Biochemistry by Lubert Stryer, 6th edition (ebook)
Part 1, Step 4 (b): 1 DNA Sequencing – Sanger’s dideoxy method Denature G G T T G G 2 A A A A Fragments of different lengths C C Electrophoresis 3 Computer Direction of migration Detector Laser 4 Action Description of the action Audio Narration (Please redraw all figures.) First show the paired strands on left top after which the arrow with ‘denature’ must appear and the strands must separate with only the ones with the colored circles remaining behind. These strands must then move into the grey tube with the label ‘electrophoresis’. Colored bands must then migrate from top to bottom of the grey tube as indicated. The cylinder marked ‘laser’ must then appear along with ‘detector’ and ‘computer’. A beam of light must then be emitted from the laser which on reaching the detector must lead to appearance of the signal on the computer screen. The synthesized strands are separated from each other, after which the differentially labelled strands of various lengths are separated by electrophoresis. The smallest fragments move further in the gel while the larger fragments remain close to the point of application. The different fluorescent labels of each ddNTP can then be detected by scanning the gel with a beam of laser. The output sequence obtained is complementary to the template strand, which can be used to deduce the original desired template sequence. As shown in animation. 5 Source: Biochemistry by Lubert Stryer, 6th edition (ebook)
Master Layout (Part 2) 1 This animation consists of 3 parts: Part 1 – Tools used for gene exploration Part 2 – Creation of a genomic library Part 3 – Transgenic plants & animals Genomic DNA 2 Splicing & ligation Vector DNA In-vitro packaging 3 Phage particles 4 Infection E. coli DNA E. coli cells Genomic library 5 Source: Biochemistry by Lubert Stryer, 5th & 6th edition (ebook)
Definitions of the components:Part 2 – Creation of a genomic library 1 1. Genomic library: A collection of host bacteria, each carrying a DNA fragment as part of a cloning vector, such that the entire set of DNA fragments when assembled represents the complete genome of an organism. 2. Genomic DNA: The entire DNA sequence of all chromosomes of an organism constitutes its genomic DNA. Genome sequencing projects aimed at deciphering the complete genome sequence of organisms including humans. 3. Splicing & ligation: The process by which the genomic DNA of interest is broken down into fragments using restriction enzymes and then inserted into a suitable vector where it is joined together or ligated using a DNA ligase enzyme. 4. Vector DNA: A vector is a DNA molecule that acts as a vehicle or medium to transfer foreign DNA into another cell. Commonly used vectors include plasmids, phages, bacterial or yeast artificial chromosomes. The DNA fragment of interest is inserted into this vector molecule by means of suitable enzymes. 5. In-vitro packaging: Process by which the vector DNA containing the genomic fragments are taken up by the phage particles and packaged such that they have a protective coating around them. 6. Phage particles: These are viruses that are capable of taking up foreign DNA and infecting bacterial cells, thereby transferring the DNA into them. 7. Infection: The process by which the phage particles attach themselves to the bacterial cells and insert their genetic material into them. 8. E. coli cells: One of the most commonly used bacterial cells for creation of genomic libraries due to their simple growth requirements and ease of manipulation. 2 3 4 5
Part 2, Step 1: 1 Restriction digestion & ligation Vector DNA Genomic DNA 2 Splicing with restriction endonucleases 3 Ligation 4 Action Description of the action Audio Narration First show the grey rectangles on top along with the colored rectangle. Next, show the arrow with label, the central part of each grey rectangle must be removed and each colored fragment must be separated and must spread out next to the grey rectangles. Nest the arrow with label must appear and the colored fragment must get inserted in between the grey rectangles with no gaps in between. Genomic DNA and DNA of the vector molecule are cleaved with the same restriction enzyme so as to generate complimentary end sequences. These are then ligated by means of the enzyme DNA ligase to generate recombinant DNA molecules. As shown in animation. 5 Source: Biochemistry by Lubert Stryer, 6th edition (ebook)
Part 2, Step 2: 1 In-vitro packaging Recombinant DNA molecules 2 3 Phage particles 4 Action Description of the action Audio Narration These recombinant molecules can be packaged in-vitro into suitable phage particles which serve as useful vectors to carry the foreign DNA molecules. Lambda and M13 are two of the most commonly used phage particles for this purpose. DNA inserts upto 10 kilobases can be inserted into these phage particles. As shown in animation. (Please redraw all figures.) Show the fragments on top as in previous slide. These must then become small and enter the figures below as shown. 5 Source: Biochemistry by Lubert Stryer, 6th edition (ebook)
Part 2, Step 3: 1 Other vector molecules EcoR1 EcoR1 Ampicillin resistance (ampR) Centromere BamH1 ori Sal I 2 Selectable marker Y Selectable marker X Pst I Tetracycline resistance (tetR) Yeast Artificial Chromosome (YAC) pBR322Plasmid 3 TEL TEL (Telomere) Origin of replication BamH1 BamH1 4 Action Description of the action Audio Narration There are other vector molecules that can take up DNA fragments from genomic DNA. Plasmids possess sites for various restriction enzymes as well as antibiotic resistance sites which help for screening purposes. Certain plasmids like pBR322 can only take up smaller DNA inserts upto ~ 10kB while larger plasmids known as bacterial artificial chromosomes can take up larger inserts upto 300 kB. The Yeast artificial chromosome is a eukaryotic vector that can take up large DNA inserts and consists of restriction sites, centromer and two telomeric sites. As shown in animation. (Please redraw all figures.) First show the circle on the left with the labels appearing sequentially in the clockwise direction starting from ‘Origin of replication’. Next, show the circle on the right with all its labels appearing sequentially starting from ‘ori’. 5 Source: Biochemistry by Lubert Stryer, 6th edition (ebook)
Part 2, Step 4: 1 Infection & amplification Infection E. coli genomic DNA E. coli cells 2 Amplification 3 Genomic library 4 Action Description of the action Audio Narration The phage molecules carrying the recombinant DNA with genomic inserts are used to infect E. coli cells. These molecules then get amplified with each round of replication of the E. coli cell. This collection of bacterial cells harboring the various genomic DNA fragments is known as the genomic library. As shown in animation. First show the ‘E.coli’ cells with the small ovals at the bottom. Next show the ‘phage particles’ getting attached to their surface. Next show the rectangular fragments inside being inserted into the rounded rectangles. These must then multiply to give several rectangles as shown below. 5 Source: Biochemistry by Lubert Stryer, 6th edition (ebook)
Part 2, Step 5: 1 Probing a genomic library for sequence of interest Radiolabelled probe Colonies adhere to paper 2 Alkali treatment Annealing with colonies of interest Cells disrupted, DNA denatured 3 Agar plate with transformed bacterial colonies Nitrocellulose sheet 4 Action Description of the action Audio Narration Once the library has been created, it is essential to have techniques to retrieve a sequence of interest. The transformed cells are selected and plated on agar, after which the plate is blotted on to a nitrocellulose paper. This is then treated with alkali, bringing about disruption of the cells and denaturation of the dsDNA. The denatured DNA is then probed for the sequence of interest by a radiolabelled molecule having a complementary sequence. As shown in animation. (Please redraw all figures.) First show the plate on left with small yellow dots. Next show the grey circle appearing on it and the dots being transferred on to the grey circle. This must move away and then be zoomed into to show the figure below. The label ‘alkali treatment’ must then appear which must make the circles porous-looking like dotted circles. Again it must be zoomed into to show the figure below. The green circles must then appear which must bind selectively to two of the grey circles as shown. 5 Source: Biochemistry by Lubert Stryer, 6th edition (ebook)
Master Layout (Part 3) 1 This animation consists of 4 parts: Part 1 – Tools used for gene exploration (Restriction enzymes, PCR, Blotting techniques) Part 2 – Creation of a genomic library Part 3 – Transgenic plants & animals Mouse without human growth hormone gene 2 Agrobacterium cell vir genes Release of cytokinins, auxins, opines Mouse with human growth hormone gene 3 T DNA Crown gall 4 Plant cell nucleus 5 Source: Biochemistry by Lubert Stryer, 5th & 6th edition (ebook)
Definitions of the components:Part 3 – Transgenic plants & animals 1 1. Ti plasmid: Tumour-inducing or Ti plasmids are plasmid molecules that are carried by the soil bacterium Agrobacterium tumefaciens. This plasmid contains genes that can bring about development of tumour state and therefore synthesis of opines. 2. T-DNA: The small 20 kb DNA insert of Ti plasmid that gets integrated into the genome of infected plant cells is known as the T-DNA. 3.Agrobacterium cell: This is a common soil bacterium known as Agrobacterium tumefaciens that is capable of introducing foreign genes into plant cells by means of its plasmid. 4. vir genes: These are the virulence genes carried by the plasmid that are essential for infection of the plant cells. 5. Crown gall: The lump of tumour tissue that is formed at the site of plant infection upon insertion of genes from the Ti plasmid by the Agrobacterium is known as the Crown gall. 6. Release of cytokines, auxins & opines: Formation of the tumour results in release of compounds such as cytokines, auxins and opines, which are metabolized by the infecting bacteria thereby disrupting the plant cell metabolism. 7. Transgenic mice: Many transgenic animals have been developed that harbour and express foreign genes of interest. The most commonly used animals are mice due to ease of handling and their convenient growth cycle. 2 3 4 5
Part 3, Step 1: 1 Functioning of Ti plasmid vir genes Agrobacterium cell Acetosyringone Release of cytokinins, auxins, opines 2 T DNA 3 Crown gall Injury to plant cell Plant cell nucleus 4 Action Description of the action Audio Narration As shown in animation. (Please redraw all figures.) First show the insides of the plant cell followed by a small tear in the outseide outline indicating ‘injury to plant’. Once this happens, the red ovals must appear out of it which must then enter the cell Once they enter the cell, the small red fragment must leave the cell and move towards the region marked ‘nucleus’,. Once it enters, the blue circles must come out of this region which must then be shown to give the figure on the right. When a plant gets injured, there is release of the phenolic compound acetosyringone, which is detected by Agrobacterium tumefaciens. Upon detection, the virulence genes of tumor-inducing (Ti) plasmid get expressed which encode the enzymes that are essential for transfer of the T DNA into the nucleus of the plant cell. Once the T-DNA gets integrated with the plant chromosome there is release of cytokinins, auxins etc. which brings about tumor formation in the plant. 5 Source: Biochemistry by A.L.Lehninger, 4th edition (ebook)
Part 3, Step 2: 1 Use of Ti plasmid for gene transfer Plating Injured leaf infected (transfer of foreign gene) Agrobacterium cell 2 Agar plate with growth hormone & kanmycin Ti plasmid without T DNA 3 Kanamycin resistance gene Growth indicating foreign gene transfer Foreign gene to be introduced 4 Action Audio Narration Description of the action As shown in animation. The useful property of infection by Agrobacterium has allowed several foreign genes of interest to be introduced into plant cells as per the requirement. One plasmid of the cell is the Ti plasmid without the T-DNA. The other plasmid contains the gene of interest along with antibiotic resistance genes placed in between two repeat units that are essential for gene transfer. The gene of interest such as genes for pesticide resistance, better yield etc. invades the plant at the site of injury. Once this happens, the foreign gene gets inserted into the plant DNA, which is confirmed by plating on to agar containing the suitable antibiotic. Only those which have taken up the gene will grow on such plates. (Please redraw all figures.) First show the ‘agrobacterium cell’ on the left with the circles in them and their labels. The circle with the red fragment must then be transferred to the leaf shown. This must then be placed in a yellow color plate as shown on right top. Once this happens, there must be growth of the plant from the green regions on the plate as shown above. 5 Source: Biochemistry by A.L.Lehninger, 4th edition (ebook)
Part 3, Step 3: 1 Transgenic animals Mice are commonly used for expression of foreign DNA. Common recombinant DNA molecules for medicine Erythropoeitin 2 Vaccines Growth factors Mouse without human growth hormone gene 3 Human growth hormone Human insulin Mouse with human growth hormone gene 4 Action Description of the action Audio Narration As shown in animation. Transgenic animals especially mice are commonly used to express foreign DNA molecules. This is often done with the help of retroviruses. Successful expression of these inserts in larger mammals has allowed the development of several recombinant products useful for medicine. (Please redraw all figures.) First show the mice as shown in image with their labels. Then show the violet box which is on top followed by each of the speech bubbles in a sequential manner. 5 Source: Biochemistry by A.L.Lehninger, 4th edition (ebook)
Interactivity option 1:Step No: 1 1 The following gel pattern is obtained after electrophoresis in the Sanger’s dideoxy method. Determine the sequence of the original template DNA molecule. Computer ddATP ddCTP 2 Direction of migration ddGTP ddTTP Detector Laser 3 c) T C C G T A T C G C T a) T G C G T A A C G C T d) A C G C A T T G C G A b) G C C G T A A C G C T 4 Results Interacativity Type Options User must be shown the animation above after which he must be given the answers to the question posed above. Once the user has viewed the animation shown above, the four answers to the question must be provided. (a) is the correct answer, which must turn green if selected by the user. All others must turn red upon selection. Choose the correct answer. 5
Questionnaire 1 1. Which of the following techniques is applicable for transfer of DNA molecules on to a nitrocellulose paper? Answers: a) Western blotting b) Northern blotting c) Southern blotting d) None of the above 2. Primer annealing takes place under which of the following conditions? Answers: a) 95oC for 1 minb) 54oC for 45 sec c) 72oC for 2 min d)80oC for 3 min 3. Which gene of the plasmid is responsible for crown gall formation? Answers:a) T-DNA b) vir genes c) Kanamycin resistant gene d) Tetracycline resistant gene 4. Which of the following enzymes is capable of elongating DNA at high temparatures? Answers: a) RNA Polymerase b) DNA Polymerase-II c) Taq Polymerase d) DNA ligase 2 3 4 5
Links for further reading Books: Biochemistry by Stryer et al., 6th edition Biochemistry by A.L.Lehninger et al., 4th edition Biochemistry by Voet & Voet, 3rd edition