DNA Recombinant Technology
DNA Recombinant Technology. DNA recombinant. Genetic Engineering. The manipulation of an organism endowment by introducing or eliminating specific gene A gene of interest is inserted into another organism, enabling it to be cloned, and thus studied more effectively


DNA Recombinant Technology
E N D
Presentation Transcript
DNA recombinant Genetic Engineering The manipulation of an organism endowment by introducing or eliminating specific gene A gene of interest is inserted into another organism, enabling it to be cloned, and thus studied more effectively Design and construction of new combinations of genes (DNA) New combinations/arrangements of DNA DNA cloning
DNA Recombinant Technology Technology used in the isolation or synthesis and joining together of unlike pieces of DNA These recombinant DNA molecules can then be introduced into bacteria, yeasts, or other cells where they can replicate and function (code for protein synthesis)
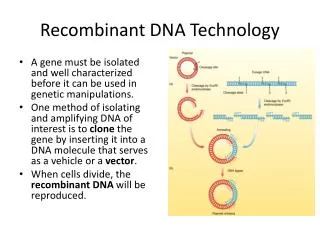
Overview of Genetic Engineering • Gene of interest is isolated from appropriate organism • Gene is recombined with a vector (carrier) DNA molecule • Recombinant DNA is introduced into appropriate host cell • Recombinant DNA is expressed at high levels in host cell • Gene product may be purified for use in treatments (antibiotics, hormones, etc.)
Why Detailed studies of the structure and function of a gene at the molecular level require large quantities of the individual gene in pure form
Cloning A collection of molecules or cells, all identical to an original molecule or cell • To "clone a gene" is to make many copies of it - for example, in a population of bacteria • Gene can be an exact copy of a natural gene • Gene can be an altered version of a natural gene • Recombinant DNA technology makes it possible
Tools Vector Restriction and ligation enzymes Host Cells
Vector • Carriers move DNA from test tubes back into cells • Pieces of DNA that can accept, carry, and replicate other pieces of DNA • An autonomously replicating genetic element used to carry DNA fragments into a host for the purpose of gene cloning • Bacterial plasmids • Bacteriophages (lambda phage) • Viruses • Yeast cells
Plasmids Naturally occurring extra-chromosomal DNA • Plasmids are circular double stranded DNA • Plasmids can be cleaved by restriction enzymes, leaving sticky ends • Artificial plasmids can be constructed by linking new DNA fragments to the sticky ends of plasmid • Maximum size of insert is about 10 kb.
Lambda • It has a genome of about 50 kb of linear DNA • Only 37 to 52 kb DNA fragments can be packaged into the lambda head. • Insertion vectors can hold up to 7 kb of cDNA. • Its life cycle is conducive to the use as a cloning vector • The lytic cycle can be supported by only a portion of the genes found in the lambda genome.
Lambda life cycle. The lytic life cycle produces phage particles immediately The lysogenic life cycle requires genes in the middle of the genome, which can be replaced
Cosmid vectors • Hybrid between a lambda vector and a plasmid. • It can contain 33 to 45 kb. Bacterial Artificial chromosomes (BAC) vectors • These vectors are based on the E. coli F factor • These vectors are maintained at 1-2 copies per cell and can hold > 300 kb of insert DNA. • Problems are low DNA yield from host cells.
Bacteriophage P1 • These vectors are like lambda and can hold up to 110 to 115 kb of DNA . • This DNA can then be packaged by the P1 phage protein coat. • The use of T4 in vitro packaging systems can enable the recovery of 122 kb inserts
Yeast Artificial Chromosomes • Many DNA fragments cannot be propagated in bacterial cells. Therefore yeast artificial chromosomes can be built with a few specific components. • Centromere • Telomere • Autonomously replicating sequence (ARS) • Genomic DNA is ligated between two telomeres and the ligation products are transformed into yeast cells
Cloning Vectors Plasmids that can be modified to carry new genes Plasmids useful as cloning vectors must have • a replicator (origin of replication) • a selectable marker (antibiotic resistance gene) • a cloning site (site where insertion of foreign DNA will not disrupt replication or inactivate essential markers
Vectors • Three important features • Cloning site • Ori-an origin of replication • A selectable marker
pBR322 The plasmid pBR322 is one of the most commonly used E.coli cloning vectors. pBR322 is 4361 bp in length and contains: (1) the replicon rep responsible for the replication of plasmid (source – plasmid pMB1); (2) rop gene coding for the Rop protein, which promotes conversion of the unstable RNA I – RNA II complex to a stable complex and serves to decrease copy number (source – plasmid pMB1); (3)bla gene, coding for beta-lactamase that confers resistance to ampicillin (source – transposon Tn3); (4) tet gene, encoding tetracycline resistance protein (source – plasmid pSC101).
pUC18/19 pUC18 and pUC19 vectors are small, high copy number, E.coli plasmids, 2686 bp in length. They are identical except that they contain multiple cloning sites (MCS) arranged in opposite orientations. pUC18/19 plasmids contain: (1) the pMB1 replicon rep responsible for the replication of plasmid (source – plasmid pBR322). The high copy number of pUC plasmids is a result of the lack of the rop gene and a single point mutation in rep of pMB1; (2) bla gene, coding for beta-lactamase that confers resistance to ampicillin (source – plasmid pBR322); (3) region of E.coli operon lac containing CAP protein binding site, promoter Plac, lac repressor binding site and 5’-terminal part of the lacZ gene encoding the N-terminal fragment of beta-galactosidase (source – M13mp18/19). This fragment, whose synthesis can be induced by IPTG, is capable of intra-allelic (alfa) complementation with a defective form of beta-galactosidase encoded by host (mutation lacZDM15). In the presence of IPTG, bacteria synthesize both fragments of the enzyme and form blue colonies on media with X-Gal. Insertion of DNA into the MCS located within the lacZ gene (codons 6-7 of lacZ are replaced by MCS) inactivates the N-terminal fragment of beta-galactosidase and abolishes alfa-complementation. Bacteria carrying recombinant plasmids therefore give rise to white colonies.
Genetic structure of the Octopine Ti plasmid Oncogenes TR TL Cyt Aux Opines
Chimeric Plasmids Named for mythological beasts with body parts from several creatures • After cleavage of a plasmid with a restriction enzyme, a foreign DNA fragment can be inserted • Ends of the plasmid/fragment are closed to form a "recombinant plasmid" • Plasmid can replicate when placed in a suitable bacterial host
Directional Cloning Often one desires to insert foreign DNA in a particular orientation • This can be done by making two cleavages with two different restriction enzymes • Construct foreign DNA with same two restriction enzymes • Foreign DNA can only be inserted in one direction
Host Cells • Propagation of a DNA sequence must take place inside a living cell (host cells) • Eschericia coli: • It provides a relatively simple and well understood genetic environment • The way to isolate plasmid is understood • It contains a single chromosome of approximately 5 Mbp • The genetic code is nearly universal • It replicates once every 22 minutes • It grows best with incubation at 37°Cin a culture medium that approximately the nutrient available in the human digestive tract
Bacterial transformation The cellular uptake and expression of DNA in a bacteria Introduction of DNA into competent cell of bacteria • Requested element in transformation: • A suitable host organism in which to insert the gene • A self-replicating vector to carry the gene into the host organism • A means of selection for host cells that have taken up the gene
Selection of Transformant A particularly important selective advantage offered by plasmid is antibiotic resistance gene It encodes for proteins that disable antibiotics secreted by microorganism with which bacteria compete Antibiotics function by several different mechanism Antibiotics resistance: A selectable marker that allows one to positively identify cells that have been induced to take up plasmid DNA Penicillin family (including ampicillin) interfere with cell wall biosynthesis Kanamycin, tetracyclin, and chloramphenicol arrest bacterial cell growth by blocking various steps in protein synthesis
Protein expression • - Gene is inserted into plasmid • Plasmid is transformed into a • host cell (E. coli) • Cell culture is prepared • Each cell contains several copies of the plasmid with gene • Gene expression leads to the production of protein • Protein level may reach 30% of • total cellular protein • Isolation of protein
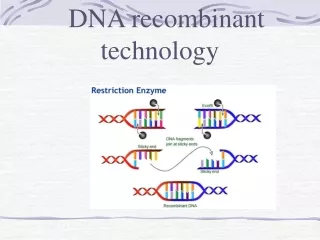
Restriction Enzymes Molecular scissors which isolated from bacteria where they are used as Bacterial defense against viruses Molecular scalpels to cut DNA in a precise and predictable manner Enzyme produced by bacteria that typically recognize specific 4-8 base pair sequences called restriction sites, and then cleave both DNA strands at this site A class of endo-nucleases that cleavage DNA after recognizing a specific sequence Members of the class of nucleases
Nuclease Breaking the phosphodiester bonds that link adjacent nucleotides in DNA and RNA molecules • Endonuclease • Cleave nucleic acids at internal position • Exonuclease • Progressively digest from the ends of the nucleic acid molecules
Restriction Enzymes • There are already more than 1200 type II enzymes isolated from prokaryotic organism • They recognize more than 130 different nucleotide sequence • They scan a DNA molecule, stopping only when it recognizes a specific sequence of nucleotides that are composed of symetrical, palindromic sequence • Palindromic sequence: • The sequence read forward on one DNA strand is identical to the sequence read in the opposite direction on the complementary strand • To Avoid confusion, restriction endo-nucleases are named according to the following nomenclature
Nomenclature • The first letter is the initial letter of the genus name of the organism from which the enzyme is isolated • The second and third letters are usually the initial letters of the organisms species name. It is written in italic • A fourth letter, if any, indicates a particular strain organism • Originally, roman numerals were meant to indicate the order in which enzymes, isolated from the same organisms and strain, are eluted from a chromatography column. More often, the roman numerals indicate the order of discovery
Restriction enzymes Restriction enzymes can be grouped by: • number of nucleotides recognized (4, 6,8 base-cutters most common) • kind of ends produced (5’ or 3’ overhang (cohesive=sticky), blunt=flush) • degenerate or specific sequences • whether cleavage occurs within the recognition sequence
A restriction enzyme (EcoRI) 1. 6-base cutter 2. Specific palindromic sequence (5’GAATTC) 3. Cuts within the recognition sequence (type II enzyme) 4. produces a 5’ overhang (sticky end)