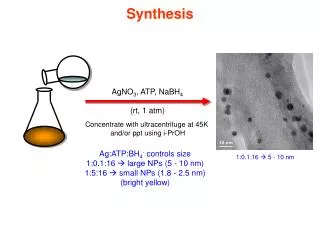
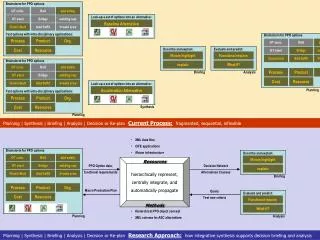
Synthesis
Synthesis. Biosynthesis. Reagents = small carbon-based molecules (e.g. amino acids, carbohydrates) Catalysts = enzymes , proteins that bind and then catalyze chemical reaction. Chapter 17: Aldehydes and Ketones – Nucleophilic Addition. 17.1 – Nomenclature of Aldehydes and Ketones.

Synthesis
E N D
Presentation Transcript
Biosynthesis Reagents = small carbon-based molecules (e.g. amino acids, carbohydrates) Catalysts = enzymes, proteins that bind and then catalyze chemical reaction
17.5 – Nucleophilic Addition Nucleophiles: H2O – hydrates H- from NaBH4 or LiAlH4 R- from RMgX, RLi NC-, ROH, RNH2, R2NH
17.6 – Hydration of Aldehydes and Ketones a Hydrate Formation (relevant to H2CrO4 oxidation) Table 17.3
17.6 – Hydration of Aldehydes and Ketones Electronic(how stable is the carbonyl – how positive is the carbon?) Steric(how crowded does the hydrate become?) Hydrate becomes more crowded, which effects the equilibrium with the precursor aldehyde or ketone
17.6 – Hydration of Aldehydes and Ketones Hydrate Formation is Base-Catalyzed Reaction faster than just with water (pH 7) Hydroxide is much more nucleophilic than water Hydroxide regenerated in second step
17.6 – Hydration of Aldehydes and Ketones Hydrate Formation is also Acid-Catalyzed Reaction faster than just H2O, H+ regenerated in second step
17.7 – Cyanohydrin Formation Both HCN and cyanide anion present in mixture Cyanide anion catalyzes the reaction, adds to the C=O New C-C bond formed (compare with Grignard)
17.8 – Acetal Formation • Acid catalyzed, use TsOH for example • Use excess ROH to drive equilibrium to the right • Water is the byproduct • Acetals are stable to bases and nucleophiles • Use excess water and H+ to go back to carbonyl - mechanism
17.8 – Acetal Formation Acetals do not react with bases or nucleophiles - useful
17.9 – Acetals as Protecting Groups Possible solution:
17.12 – The Wittig Reaction Phosphonium salt Wittig ylide
17.12 – The Wittig Reaction Retrosynthesis: Synthesis: