Design and Synthesis of Tic Peptidomimetics Mimicking Reverse Turn Secondary Structures
Explore Tic-based structures mimicking reverse turn motifs using computational and synthetic methods, with emphasis on stereochemistry and stable conformations.

Design and Synthesis of Tic Peptidomimetics Mimicking Reverse Turn Secondary Structures
E N D
Presentation Transcript
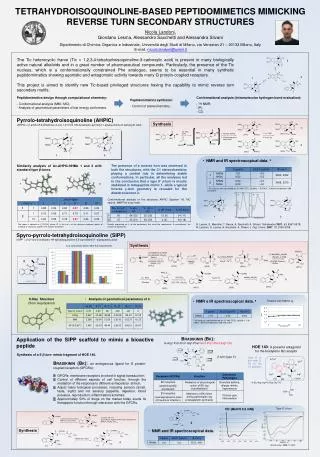
Tetrahydroisoquinoline-based peptidomimetics mimicking reverse turn secondary structures Nicola Landoni, Giordano Lesma, Alessandro Sacchetti and Alessandra Silvani Dipartimento di Chimica Organica e Industriale, Università degli Studi di Milano, via Venezian 21 – 20133 Milano, Italy E-mail: nicola.landoni@unimi.it The Tic heterocyclic frame (Tic = 1,2,3,4-tetrahydroisoquinoline-3-carboxylic acid) is present in many biologically active natural alkaloids and in a great number of pharmaceutical compounds. Particularly, the presence of the Tic nucleus, which is a conformationally constrained Phe analogue, seems to be essential in many synthetic peptidomimetics showing agonistic and antagonistic activity towards many G protein-coupled receptors. This project is aimed to identify new Tic-based privileged structures having the capability to mimic reverse turn secondary motifs. 2 3 1 Peptidomimetics design through computational chemistry: - Conformational analysis (MM / MC);- Analysis of geometrical parameters of low energy conformers. Conformational analysis (intramolecolar hydrogen bond evaluation): - 1H NMR;- IR;- CD. Peptidomimetics synthesis: - Control of stereochemistry . Pyrrolo-tetrahydroisoquinoline (AHPIC)(AHPIC = 2-amino-8,9-dimethoxy-3-oxo-1,2,3,5,6,10b-hexahydro-pyrrolo[2,1-a]isoquinoline-5-carboxylic acid) Synthesis 1 2 1 2 • NMR and IR spectroscopical data. a The presence of a reverse turn was observed in both the structures, with the C1 stereochemistry playing a central role in determining stable conformations. In particular, all the analyses led to the conclusion that a type II’ b-turn is mostly stabilized in tetrapeptide mimic 1, while a typical inverse γ-turn geometry is revealed for the diastereoisomer 2. Similarity analysis of Ac-AHPIC-NHMe 1 and 2 with standard type β-turns. 1 2 aAll analyses were performed on 3.0 mM CDCl3 solutions. b At 298 K. c Determined between 298 and 328 K. Conformational analysis of the structures AHPIC (Spartan ‘06, MC search, MMFF94 force field): 1 2 G. Lesma, E. Meschini, T. Recca, A. Sacchetti, A. Silvani, Tetrahedron 2007, 63, 5567-5578; N. Landoni, G. Lesma, A. Sacchetti,A. Silvani, J. Org. Chem. 2007, 72, 9765-9768. The score is defined as [(1-R2)/N], where R2 is the r.m.s. of the distances between points of similarity of structures and N is the number of centres. Results expressed as % of the conformers that meet the requirement. In parentheses, the number of conformers. Spyro-pyrrolo-tetrahydroisoquinoline (SIPP)(SIPP = 2-(2'-oxo-2,4-dihydro-1H-spiro[isoquinoline-3,3'-pyrrolidine]-1'-yl)propanoic acid) Synthesis % of conformers which meet the requirements. 3 1 3 • Analysis of geometrical parameters of 3. X-Ray Structure (from isopropanol) • NMR e IR spectroscopical data. a Titration with DMSO-d6 aAll analyses were performed on 3.0 mM CDCl3 solutions. b At 298 K. c Determined between 298 and 328 K. Bradikinin (Bk): H-Arg1-Pro2-Pro3-Gly4-Phe5-Ser6-Pro7-Phe8-Arg9-OH Applicationof the SIPP scaffoldtomimic a bioactive peptide HOE 140: A powerful antagonist for the bradykinin B2 receptor Synthesis of a II’b-turn mimic fragment of HOE 140. b-turn (type II’) Type II’ b-turn with D-Tic in the i+1 position. • Bradikinin (Bk):an endogenous ligand for G protein-coupled receptors (GPCRs): • GPCRs: membrane receptors involved in signal transduction; • Control of different aspects of cell function, through the mediation of the response to different extracellular stimuli; • Adjust many biological processes, including sensory (smell, taste, sight) and not sensory (appetite, digestion, blood pressure, reproduction, inflammation) activities; • Approximately 50% of drugs on the market today exerts its therapeutic function through interaction with the GPCRs. Type II’ b-turn CD (MeOH 0.2 mM) 4 Synthesis • NMR and IR spectroscopical data. 4 Biochemistry, 1978, 17, 4951.