Understanding the Classification of Matter
90 likes | 221 Vues
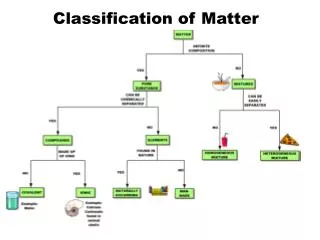
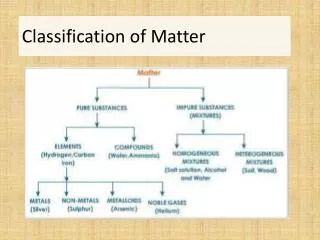
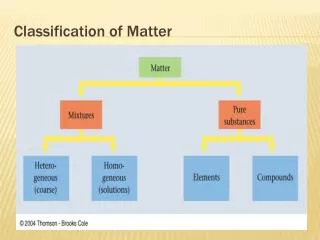
Matter is everything that has mass and takes up space. It can be classified into two main types: substances and mixtures. Substances are pure and have a consistent chemical composition, made up of elements (one type of atom) and compounds (two or more atoms chemically bound). Mixtures consist of two or more substances physically combined, which can be homogeneous (uniform throughout, like solutions) or heterogeneous (varying composition, like cereal and milk). Understanding these classifications helps us grasp the nature of the materials around us.

Understanding the Classification of Matter
E N D
Presentation Transcript
Matter • Has mass • Takes up space • Everything is made up of it
Substance • Same chemical composition; pure. They stay the same all the way through; they are made up of elements and compounds • Elements- pure substances that are made up of one type of atom; atoms are the smallest part in an elements • Compounds- are 2 or more elements that are chemically bound; molecules are the smallest piece of a compound
Mixture • When two or more things are physically put together They can be . . • homogeneous (solution)- they are the same through out, when something dissolves into something ex: Kool Aid, lemonade • Heterogeneous- they have compositions that vary ex: cereal and milk
Heterogeneous • Are made up of suspensions and colloids • Suspension- are particles that are big enough to be seen and they settle out • Colloid- are in a gel, foam, or cream form. The particles do not settle but they are larger than the solution
1) What are the two types of matter? a) Substances & Mixtures b) Compounds & Solutions c) Molecules & Colloids 2) Which are examples of colloids a) water, air b) gold, sliver c) gel, foams and creams Questions (:
3)What are substances made of? a) suspensions & colloids b) mixtures & compounds c) elements & compounds 4) What is the smallest part of a compound? Atom Solution Molecule Questions
Questions 5)What has mass? • Only solids • Everything • Only elements